Jargon is something we should work hard to avoid, and yet there is an awful lot of it we find difficult to let go. Right at the top is the GxPs.

GxP is a general abbreviation for the “good practice” quality guidelines and regulations. The “x” stands for the various fields, including the pharmaceutical and food industries, for example good manufacutiring practice, or GMP.
There are a lot of GxPs, though we tend to focus on 5(ish), depending on where you are.

We tend to argue a lot about them. Even to the GxP vs GXP. Or GPvP vs GVP. Or GdocP or GDP (so damn confusing, there is another GDP – Good Distribution Practices). Or if Good Storage Practice is its own body or part of the GMPs and GDPs. And…and…and.. The arguing can be fun.
The Five big ones in pharma and medical devices are GLP, GCP, GMP, GDP and GPvP. Some of the others like GACP are pretty intesting in their application.
Some like GDocP and GAMP are more specific threads that go across the GxPs.
By nature the GxPs are tied to the phase of the pharmaceutical pipeline.
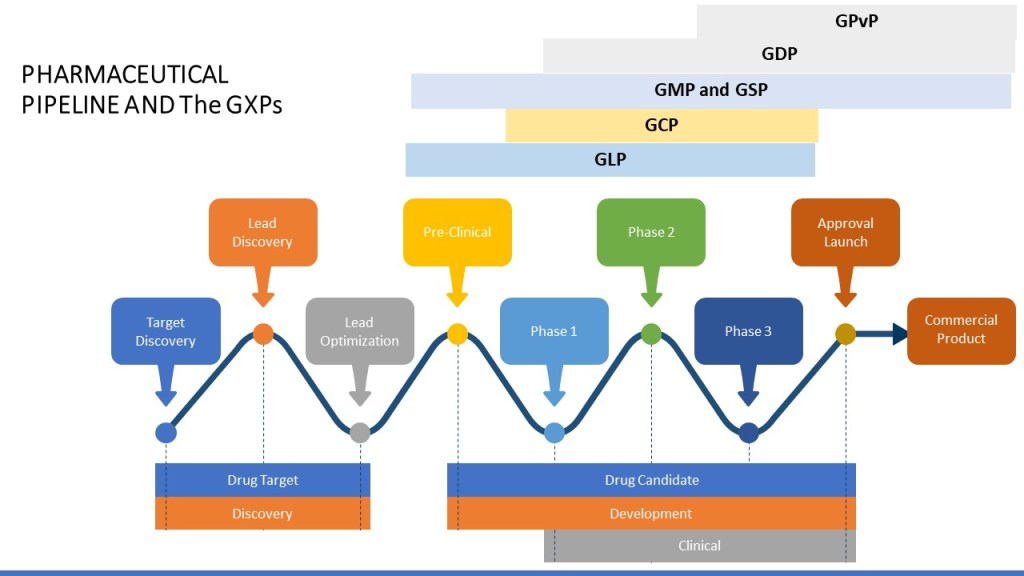
The GxPs are all about ensuring compliance and are informed from a wide range of sources, starting with law and regulations.
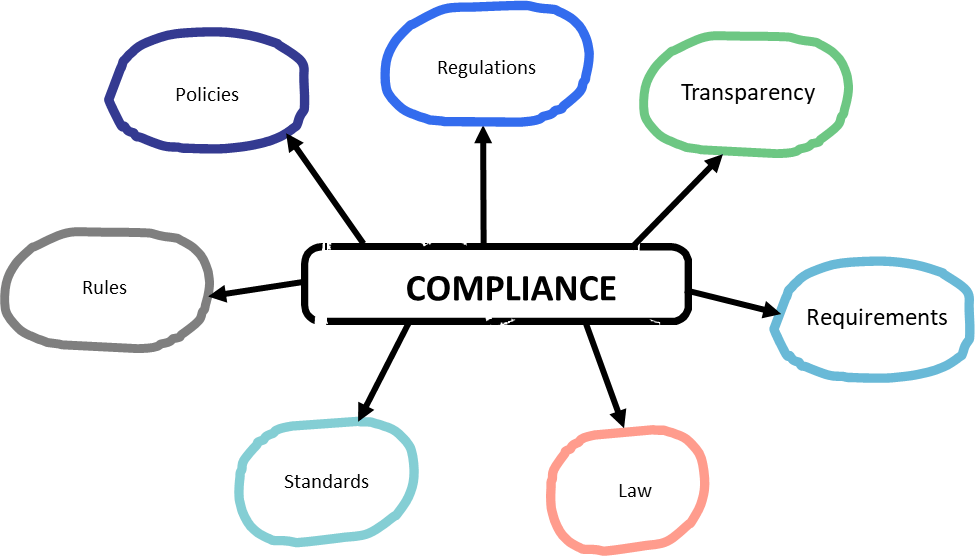
Being in the age of globalization, there are many many sources to draw from.

This can also draw from beyond the health authorities (for example in the US USDA for GACP or the DEA for parts of the GDPs).
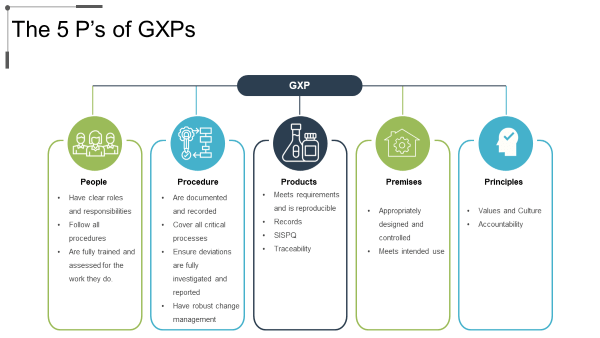
At the end of the day, GxPs answer to five important criteria.

5 thoughts on “The GxPs – a brief definition”