Change Management and Change Control is a topic that I cannot discuss enough. Which is a good thing because I am presenting on it at the 2018 ASQ World Conference.
Below is the proposal I submitted.
Session Type: Workshop
Title: Change Mgmt & Control-Lessons from Consent Decree
Presentation Description:
Drawing on the experiences of a company under a FDA consent decree, this workshop will consider change management and change control, and how they are improved upon by a grounding in knowledge management and risk management. The lessons learned and applied are relevant not only to the pharmaceutical industry, but to other FDA industries (medical devices, cosmetics, food, etc.), and to other heavily regulated industries (energy, mining, financial services and beyond). It is easy for a company in these situations to focus on change control and forget the wider scope of change management. During this workshop we will share improvements made in our processes as a result of being under the consent decree, as well as several templates, and tools to enable good change management, and change control activities.
Presentation Summary:
The Pharmaceutical Quality System described by the International Conference of Harmonization (ICH) is a holistic approach which facilitates the consistent development and production of high quality pharmaceutical products. It aims to support innovation and continual improvement of products, processes, and methodologies using knowledge management and quality risk management. Providing a lifecycle approach to pharmaceuticals, change management is a key element to this approach.
The Pharmaceutical Industry is one of the most regulated industries. In the US, the Food and Drug Administration (FDA) often uses the consent decree as the ultimate enforcement tool for those who break the rules. A company under consent decree needs to prove, via third party, that it has achieved and can sustain regulatory compliance. Companies under a consent decree must break down and build up their entire quality system.
After this workshop attendees will be able to:
- Evaluate lessons learned from a consent decree and building a robust pharmaceutical quality system for your use.
- Explain how the pharmaceutical experience can deepen your understanding of ISO 9001:2015, especially risk based thinking
- Identify how change management fits into a culture of quality
- Understand how the change management system increases compliance
- Use a few simple but effective tools for change management.
The ICH quality approach developed from ISO 9001:2008 but in many ways preceded the risk based thinking pivotal to IS 9001:2015. Utilizing a matrix of similarities and differences between traditional GMPs, the ICHs and ISO 9001:2015, we will explore several lessons learned from consent decree activities, and demonstrate principles of change management.
This workshop will explore change management from the three lenses of science, regulation and risk, with a focus on knowledge management and risk management as enablers of successful change management. Several examples will be shared to demonstrate the fundamental connections between these systems.
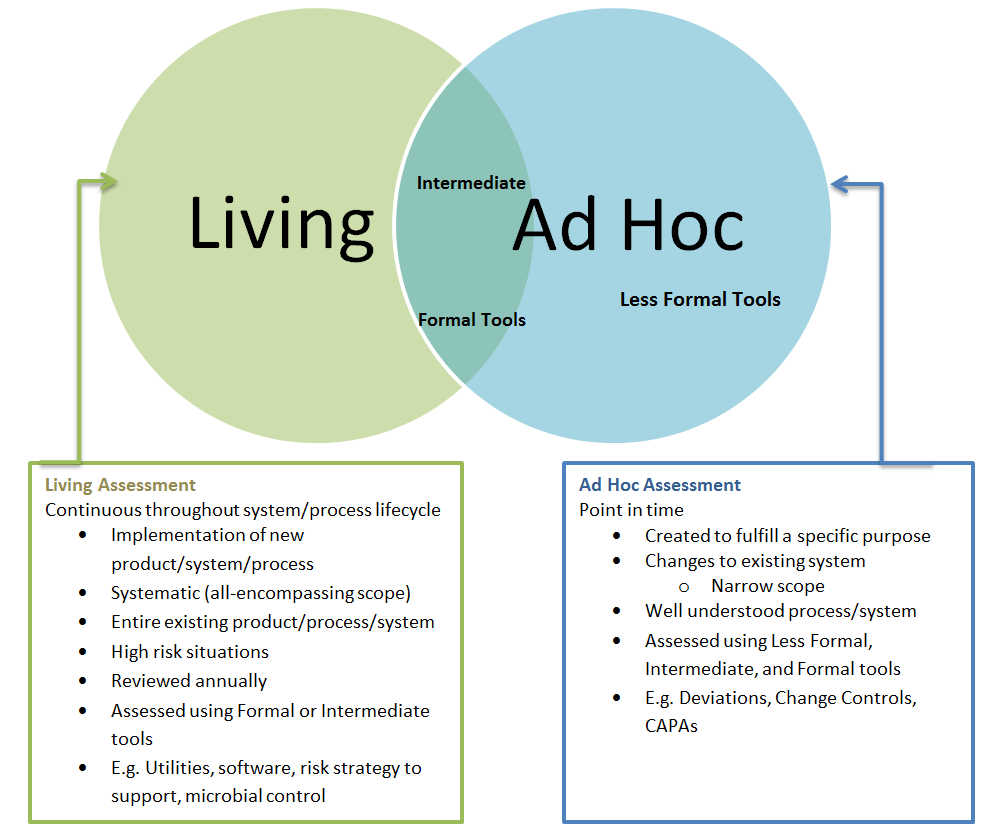
The basis of this workshop will be change management as a fundamental part of a culture of quality. We will share best practices based on lessons learned from the consent decree around the impact of cumulative changes and large-scale versus incremental change.
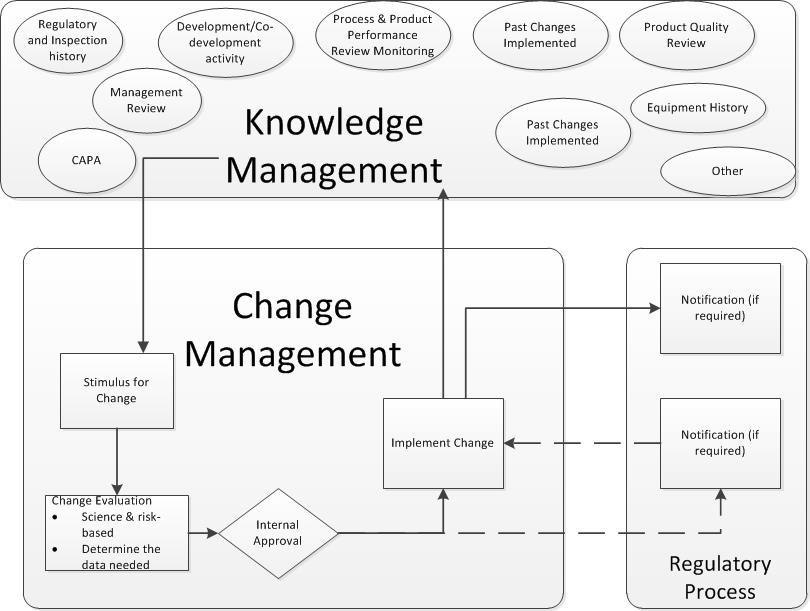
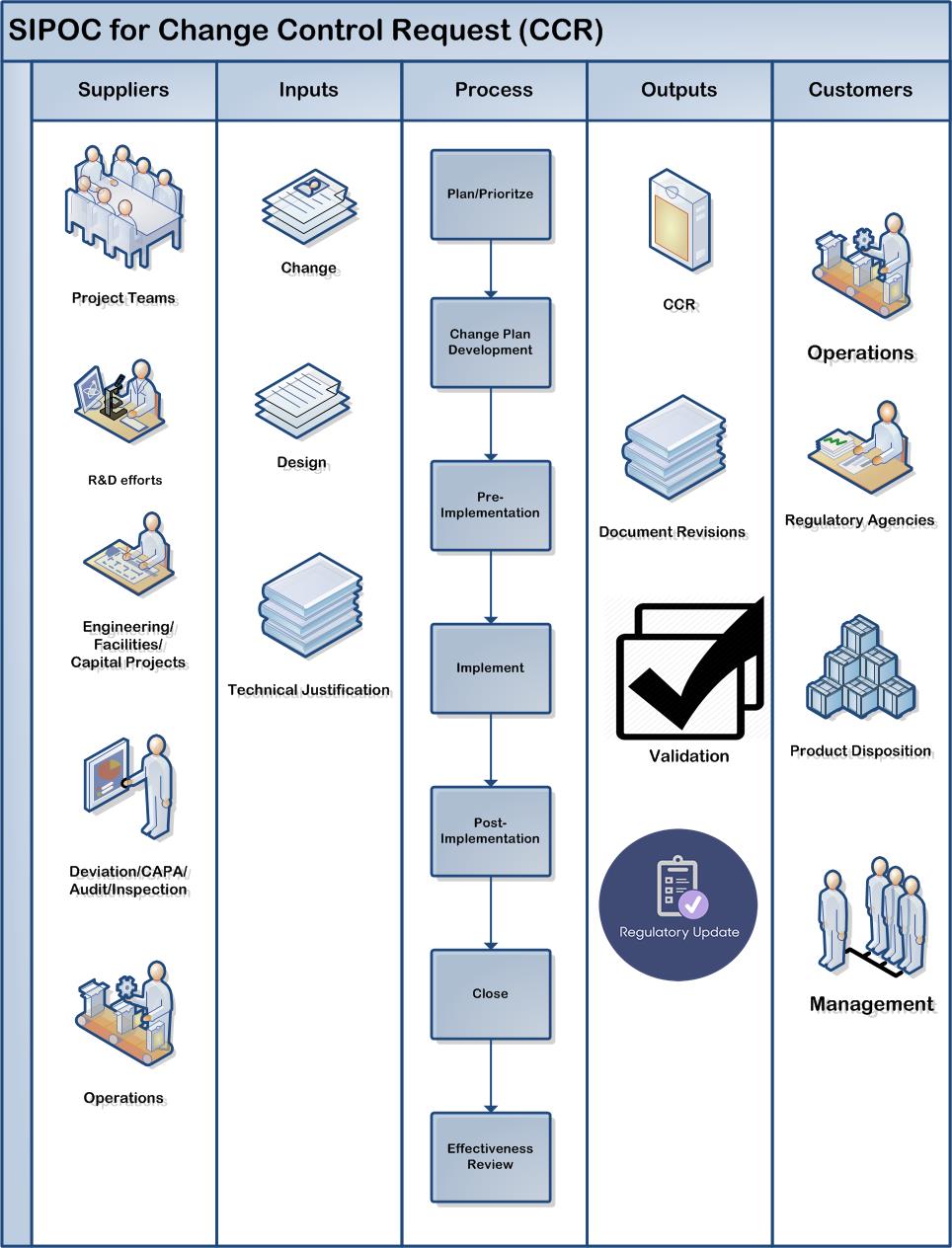
Compliance for change management necessitates clear accountabilities, prioritization of changes, and the role of the quality unit – including the importance of a change champion or steward. This workshop will explore the relationship between change control, which often refers to the execution step of an individual change, and change management, which is a more systematic, holistic approach to the review and management of a portfolio of changes and the change process.
Managing change on the personnel and organization will be evaluated, especially as it interfaces with knowledge management. Several tools will be shared for determining and mitigating risk and impact, including a training matrix.
By the end of the workshop we will have shared several tools, including a matrix to evaluate cumulative changes, a risk-based change evaluation tool, and a scope setting method that incorporates an evaluation of knowledge and risk.
Focus Area: Risk and Change
Presentation level: Intermediate
Industry Area: Food/Drug/Cosmetic, but all manufacturing can benefit
Presentation Best Matches this Technical Area: Quality Management Principles
Who should attend: Senior/Executive Officer, Director, Manager, Supervisor, and all others involved in change management
Presenter information: Jeremiah Genest, CMQ/OE, CPGP. Sanofi – Genzyme, Quality Systems Manager
Biography: Jeremiah Genest has 20 years quality systems experience in the energy and pharmaceutical industry, with over a decade’s experience implementing and running quality systems in the biotech field. For the last four years, he has managed change management at Sanofi’s Allston Landing Facility, which is under an FDA Consent Decree. He is a Certified Manager of Quality/Operation Excellence and Pharmaceutical GMP Professional.