Theresa Mullin, FDA’s Associate Director for Strategic Initiatives for the Center for Drug Evaluation and Research recently gave a presentation “Update from FDA CDER” at GMP by the Sea (I need to go to that that some-year).
As in other FDA presentations this presentation summarized the Quality Metrics Research Final Report by the University of St. Gallen as the appropriate steps to ensure quality maturity:
- Optimized set-up and cleaning procedures are documented as best practice process and rolled out throughout the whole plant.
- A large percentage of equipment on the shop floor is currently under statistical process control.
- For root cause analysis, the firm has standardized tools to get a deeper understanding of the influencing factors for problems.
- Goals and objectives of the manufacturing unit are closely linked and consistent with corporate objectives and the site has a clear focus.
- Manufacturers have joint improvement programs with suppliers to increase performance.
- All potential bottleneck machines are identified and supplied with additional spare parts.
- For product and process transfers between different units or sites,standardized procedures exist that ensure a fast, stable and compliant knowledge transfer.
- Charts showing the current performance status such as current scrap rates and current up times are posted on the shop floor and visible for everyone.
- The firm regularly surveys customers’ requirements.
- The firm ranks its suppliers and conducts supplier qualifications and audits.
This are some pretty low hanging fruit. They are also the pretty necessary in any organization, not just pharmaceuticals.
There was also a little discussion on the use of Q10 that really makes me wish I had been there to hear exactly what was said. I hope it was “Just freaking implement it already.”
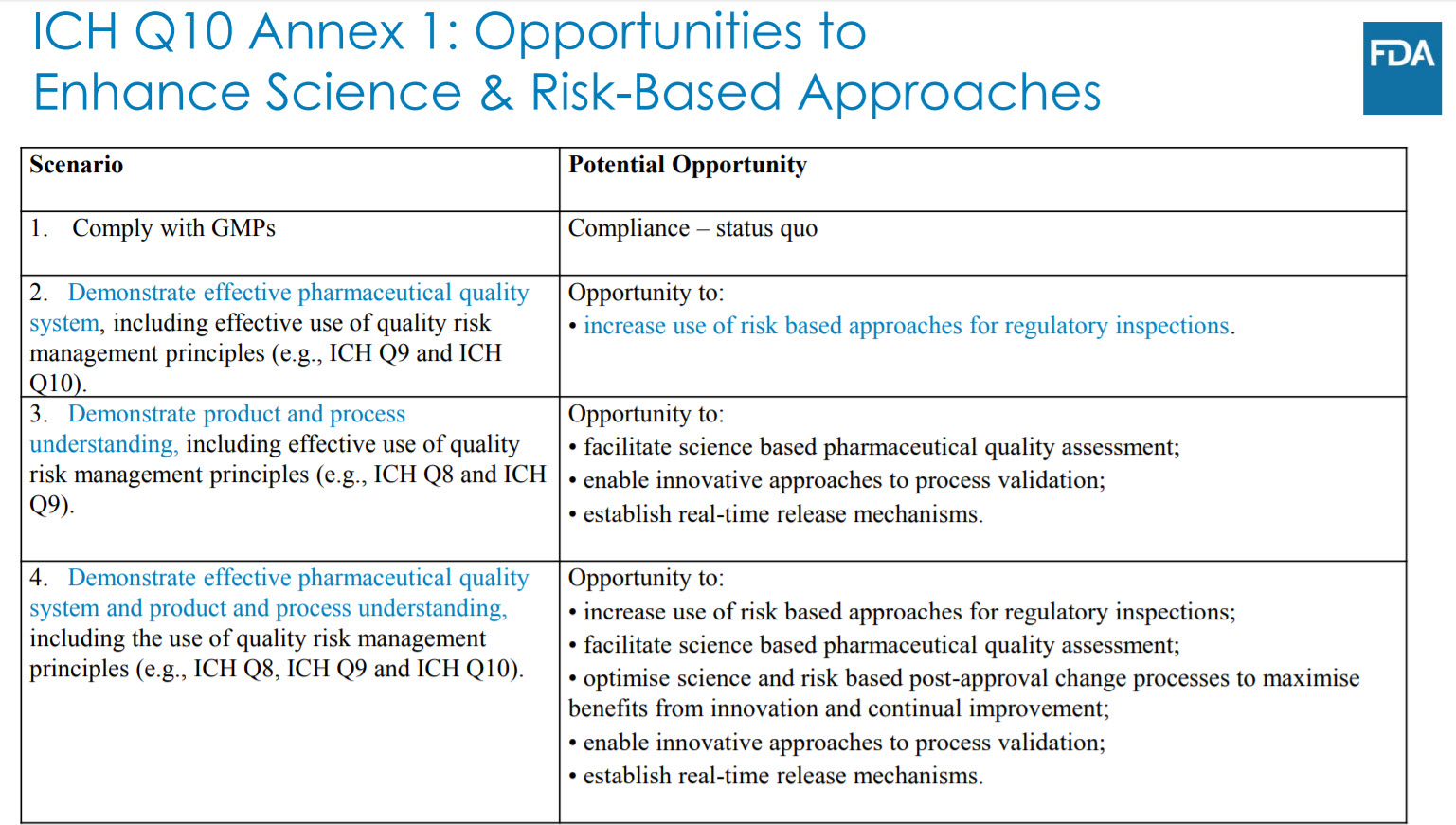
In general, useful slides, I recommend going and checking them out.
