Google does not share search terms for privacy reasons, so it is rare that a search term stands out on my blog.
So here you go.
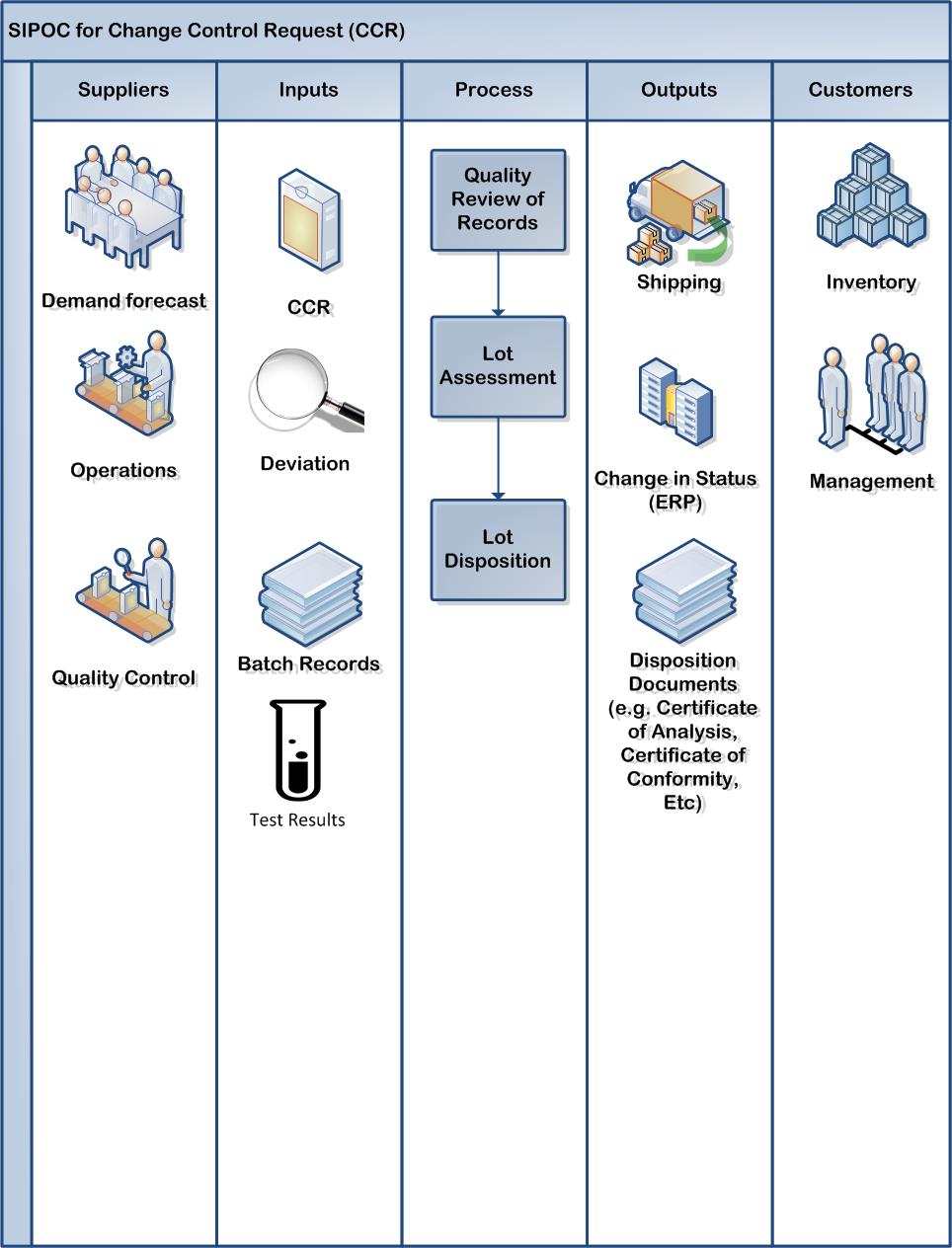
This is purposefully a high level process.
Quality Review of Records: Batch Record, Packaging Record, the like
Lot Assessment: Evaluation of deviations, of change control and test results; but also of other inputs such as Critical Utilities and Environmental Monitoring Review. Ideally a holistic view.
Lot Disposition: Decision that the product meets all requirements of the GMPs and the market authorization.
Some important regulatory requirements:
- United States: 21 CFR 211.22(a); 211.22(d)
- EU: 1.4(xv); 1.9(vii); EU Annex 16
- World Health Organization: Annex 3-GMP 1.2(g); Annex 3-GMP 9.11, 9.13, 9.15
One of the drivers for digital transformation, and a concept at route of the ICHs, is the idea of release by exception. Our systems will be tight enough, our design space robust enough, that most products are automatically released and sent into the market.
