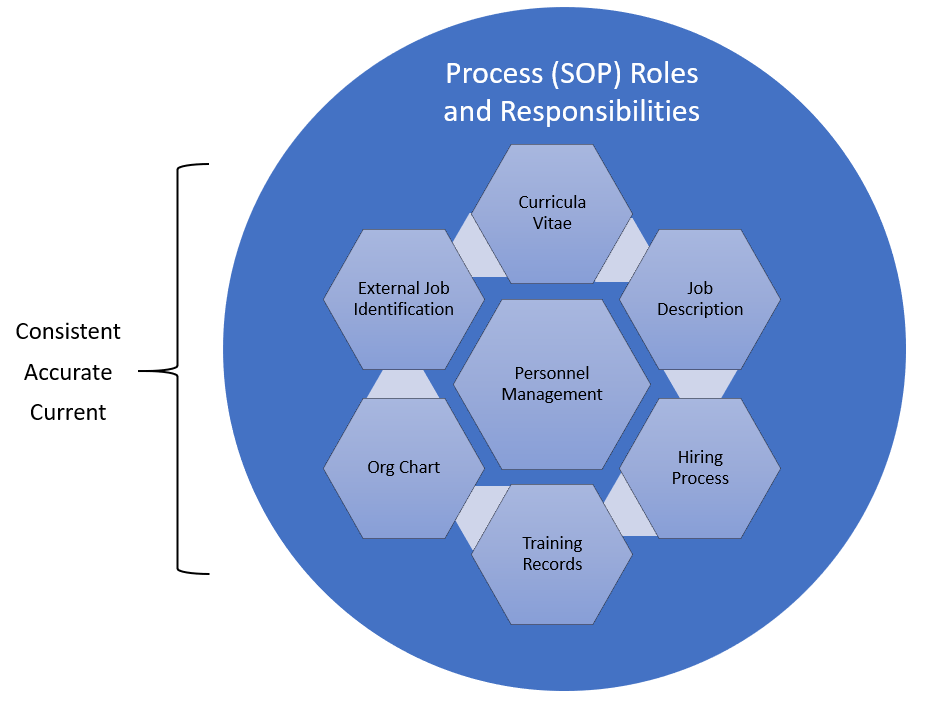
In the post “HR and Quality, joined at the hip” I covered some of the regulations that set the expectations in the pharmaceutical and medical device industries that employees have the appropriate education, experience and training to do their jobs. What is often called the personnel qualification triangle.
A job description is a written document that outlines the duties, responsibilities, contributions, behaviors, outcomes and required qualifications for a specific job in an organization. A good job description is a specification that results from a detailed job analysis. It is used in hiring and performance evaluation. It is also the starting point for determining a good GxP training plan.
In order to providing the specific knowledge, skills, abilities and behaviors that need to be addressed for each employee, the job description needs to specifically call out the roles in the processes an employee will play. Instead of providing broad statements like “participate in CAPAs” or “Manufacture product” it should be more specific such as “create and project manage CAPAs” or “Perform visual inspection.”
I challenge everyone to think broadly about the job description as a tool to drive excellence. Utilized throughout the life of employment, a job description is a powerful tool that can aid managers. Managers have a road map that can help them with their duties of planning, leading, organizing, controlling and staffing. With a road map, the complexities of the organization become easier.
The curricula vitae provides evidence that the employee is fit-for-purpose to the job description. The curricula vitae shows education and experience that demonstrates the possession of knowledge, skills, abilities and behaviors.
The training plan then lays out what the employee needs to bit fit-for-use, to be able to do all the roles in the job description. It includes all the process and role specific training, as well as filling in any gaps that might exist on the curricula vitae.
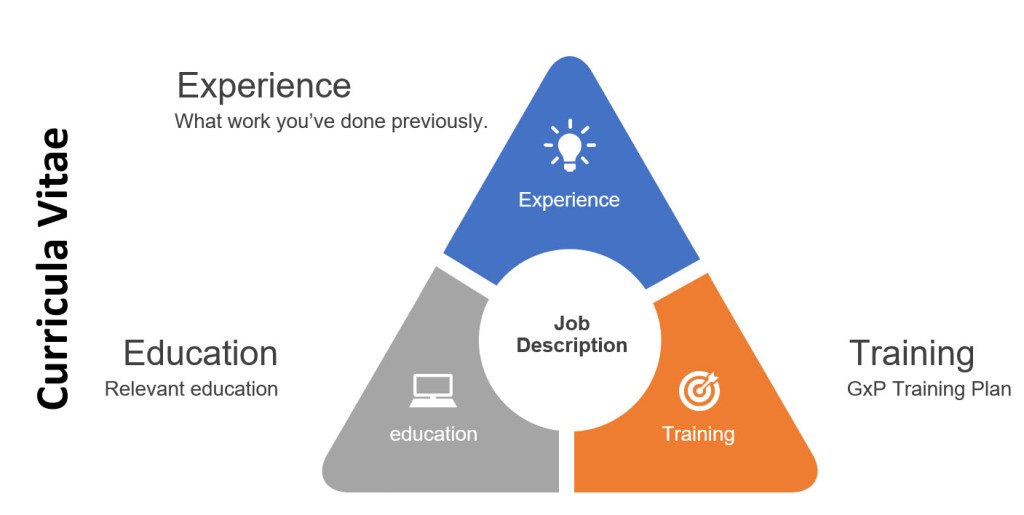
It is important to note that this may not be a fine equilateral triangle. Experience, for example, can often, but not always make up for education.
Failure Points in the Personnel Qualification Triangle
- Position and experience descriptions on CVs do not match the corresponding job description. This red flag stems from a lack of coordination between the curricula vitae and job description, which can be particularly concerning when an employee has a job description that requires very specific technical knowledge or oversees other seemingly unrelated areas that their experience would indicate.
- Employee positions current position is not included in curricula vitae. The curricula vitae should always include the current position. While not a deal breaker, this is perhaps the easiest way to see large gaps in the cv, especially if the employee moves around or up in the same organization.
- Curricula vitae do not reflect the level of experience expected given the employee’s job title.
- The roles and responsibilities documented in in the job descriptions do not correspond with those included in SOPs. A reviewer should be able to go from a process to anyone engaged in the process and be able to see the work the individual does reflected.
- Job titles match. Curricula vitae, job descriptions and what an individual is listed as on an organizational chart need to all match. I usually go as far to check someone’s business card before they go into meet with an inspector or external auditor.