The interface between the Quality and Human Resources departments in pharma and medical devices can be poorly understood by many leaders in both departments. Quality tends to focus on product and process, HR on hiring, benefits, stuff like that. As a quality professional who oversees the training and personnel qualification system, I tend to sit between the two.
A quick summary of some regulations are in order. This is by no ways a comprehensive list.
| Regulation | Requirement |
| ICH E6 R2, 2.8 | Each individual involved in conducting a trial should be qualified by education, training, and experience to perform his or her respective task(s). |
| US FDA 21CFR 211.25 | (a) Each person engaged in the manufacture, processing, packing, or holding of a drug product shall have education, training, and experience, or any combination thereof, to enable that person to perform the assigned functions. Training shall be in the particular operations that the employee performs and in current good manufacturing practice (including the current good manufacturing practice regulations in this chapter and written procedures required by these regulations) as they relate to the employee’s functions. Training in current good manufacturing practice shall be conducted by qualified individuals on a continuing basis and with sufficient frequency to assure that employees remain familiar with CGMP requirements applicable to them. (b) Each person responsible for supervising the manufacture, processing, packing, or holding of a drug product shall have the education, training, and experience, or any combination thereof, to perform assigned functions in such a manner as to provide assurance that the drug product has the safety, identity, strength, quality, and purity that it purports or is represented to possess. (c) There shall be an adequate number of qualified personnel to perform and supervise the manufacture, processing, packing, or holding of each drug product. |
| Canada C.02.006 | Every lot or batch of a drug shall be fabricated, packaged/labelled, tested and stored under the supervision of personnel who, having regard to the duties and responsibilities involved, have had such technical, academic, and other training as the Minister considers satisfactory in the interests of the health of the consumer or purchaser. |
| EU EMA/INS/GMP/735037/201 2.1 | All parts of the Pharmaceutical Quality system should be adequately resourced with competent personnel, and suitable and sufficient premises, equipment and facilities. |
| WHO Annex 3-GMP | 9.2 The manufacturer should have an adequate number of personnel with the necessary qualifications and practical experience. The responsibilities placed on any one individual should not be so extensive so as to present any risk to quality. 9.3 Responsible staff should have its specific duties recorded in written descriptions and adequate authority to carry out its responsibilities. Its duties may be delegated to designated deputies of a satisfactory qualification level. There should be no gaps or unexplained overlaps in the responsibilities of personnel concerned with the application of GMP. The manufacturer should have an organization chart. (also see section 9.6 and 9.7 on key personnel) |
| WHO Annex 5-GDP 7.2 | Key personnel involved in the distribution of pharmaceutical products should have the ability and experience appropriate to their responsibility for ensuring that pharmaceutical products are distributed properly |
| Guideline on good pharmacovigilance practices (GVP) EMA/541760/2011 | Achieving the required quality for the conduct of pharmacovigilance processes and their outcomes by an organisation is intrinsically linked with the availability of a sufficient number of competent and appropriately qualified and trained personnel (see I.B.6.). All personnel involved in the performance of pharmacovigilance activities shall receive initial and continued training [IR Art 10(3), Art 14(2)]. For marketing authorisation holders, this training shall relate to the roles and responsibilities of the personnel [IR Art 10(3)]. |
| 21 CFR 58.29 | (a) Each individual engaged in the conduct of or responsible for the supervision of a nonclinical laboratory study shall have education, training, and experience, or combination thereof, to enable that individual to perform the assigned functions. (b) Each testing facility shall maintain a current summary of training and experience and job description for each individual engaged in or supervising the conduct of a nonclinical laboratory study. (c) There shall be a sufficient number of personnel for the timely and proper conduct of the study according to the protocol. |
| 21CFR 820.25(a) | Each manufacturer shall have sufficient personnel with the necessary education, background, training, and experience to assure that all activities required by this part are correctly performed. |
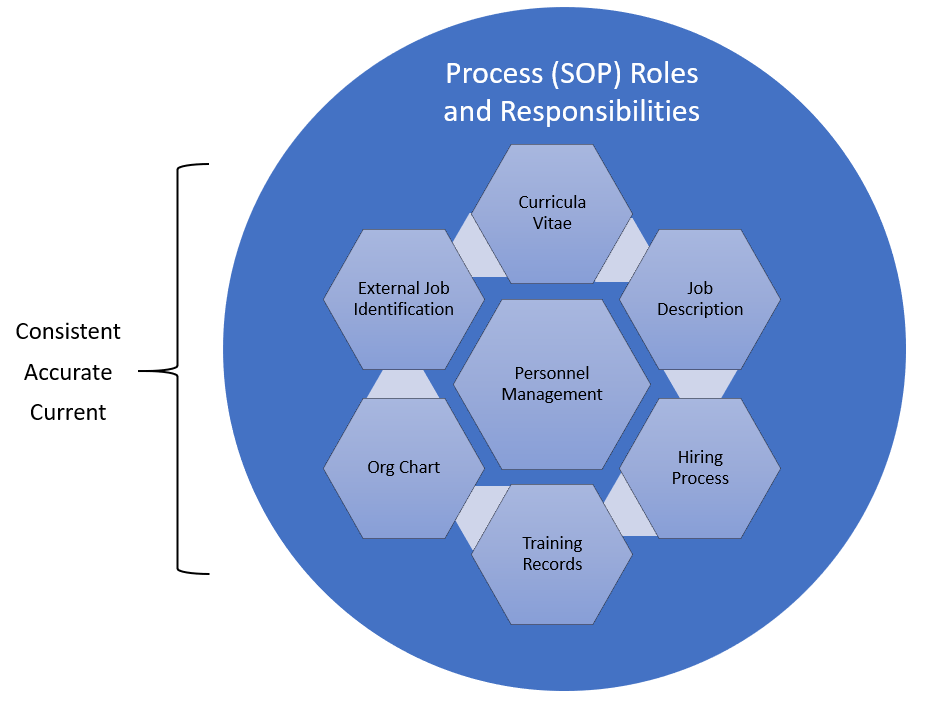
This assortment of regulations provides structure for every aspect of employment from how we hire to how we manage people. We can divide this into the following major areas: Curricula Vitae, Job Description, Hiring Process, Training Records, Org Chart, External Job Identification. All informed by the rest of our quality system, especially Process (SOP) Roles and Responsibilities.
Like most things that concern us we want all of this to be consistent and accurate.
Looking at the 2020 FDA 483 data we can see that these regulations are a concern throughout organizations.
| Citation Program Area | Reference Number | Short Description | Long Description | Frequency |
| Bioresearch Monitoring | 21 CFR 58.29(a) | Personnel: education, training, experience | Not all individuals engaged in the conduct of or responsible for the supervision of a nonclinical laboratory study have education, training, and experience, or combination thereof, to enable that individual to perform assigned functions. Specifically, *** | 1 |
| Devices | 21 CFR 820.25(b) | Training – Lack of or inadequate procedures | Procedures for training and identifying training needs have not been [adequately] established. Specifically, *** | 30 |
| Devices | 21 CFR 820.25(b) | Training records | Personnel training is not documented. Specifically, *** | 18 |
| Drugs | 21 CFR 211.25(a) | Training–operations, GMPs, written procedures | Employees are not given training in [the particular operations they perform as part of their function] [current good manufacturing practices] [written procedures required by current good manufacturing practice regulations]. Specifically, *** | 18 |
| Drugs | 21 CFR 211.25(a) | Training , Education , Experience overall | Employees engaged in the [manufacture] [processing] [packing] [holding] of a drug product lack the [education] [training] [experience] required to perform their assigned functions. Specifically, *** | 14 |
| Drugs | 21 CFR 211.25(a) | GMP Training Frequency | GMP training is not conducted [on a continuing basis] [with sufficient frequency] to assure that employees remain familiar with CGMP requirements applicable to them. Specifically, *** | 7 |
| Drugs | 21 CFR 211.25(b) | Supervisor Training/Education/Experience | Individuals responsible for supervising the [manufacture] [processing] [packing] [holding] of a drug product lack the [education] [training] [experience] to perform their assigned functions in such a manner as to assure the drug product has the safety, identity, strength, quality and purity that it purports or is represented to possess. Specifically, *** | 2 |
Reaching beyond the regulations, we really need to ensure that a fear climate does not exist inside the organization, what is often called psychological safety. Looking to Deming, quality should extend to the performance check processes, and frankly those that introduce ranking of employees or departments are not the best for a culture of excellence.
There are many transactional differences in the roles, but if quality is not at the table for key decisions on hiring and culture the organization has missed opportunities.

14 thoughts on “HR and Quality, joined at the hip”