Last November, officials from the European Medicines Agency (EMA) and US Food and Drug Administration (FDA) met with industry representatives in London to discuss the various quality challenges that arise when the development of investigational products is accelerated .
The report was recently published, and can be found here.
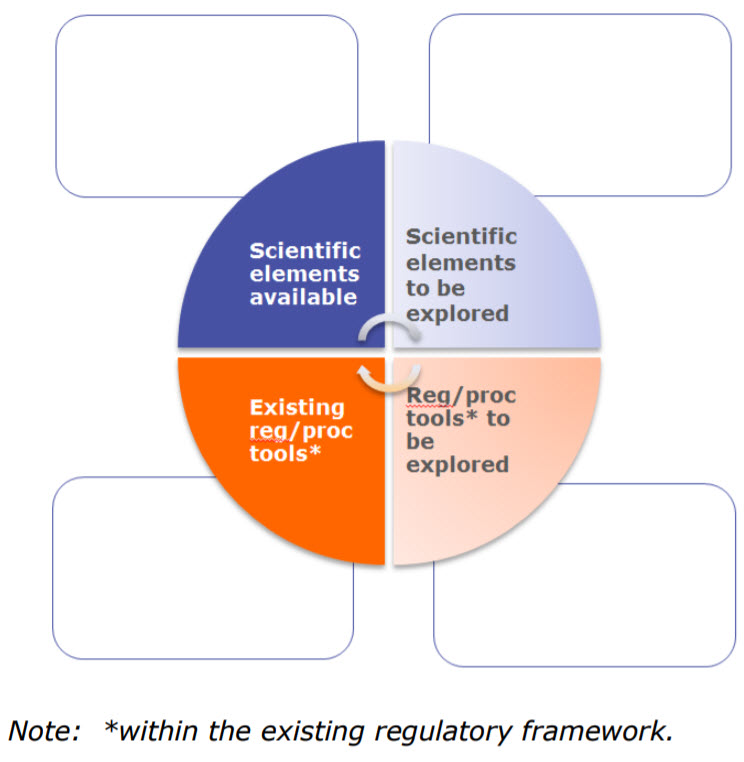
The workshop discussed process validation, control strategies, good manufacturing practice (GMP) compliance, comparability, stability and regulatory tools of early access approaches. Throughout they discussed two elements:
- Scientific which includes technologies and scientific concepts or principles for development, manufacture and quality risk management, which may or not be present or implied in existing guidelines. Examples include concurrent validation, new modelling methodologies, new analytical techniques, etc.
- Regulatory/procedural tools are described in the legal, regulatory framework and can be specific to PRIME (or Breakthrough Therapies) (e.g. kick-off meetings) or generally applicable [e.g. Post-approval change management protocols (PACMPs), recommendations, scientific advice (SA)].
I strongly recommend reading the report in it’s entirety.

One thought on “Quality Challenges of Accelerating Investigational Products”