The biotech industry is experiencing a significant transformation in validation processes, driven by rapid technological advancements, evolving regulatory standards, and the development of novel therapies.
The 2024 State of Validation report, authored by Jonathan Kay and funded by Kneat, provides a overview of trends and challenges in the validation industry. Here are some of the key findings:
- Compliance and efficiency are top priorities: Creating process efficiencies and ensuring audit readiness have become the primary goals for validation programs.
- Compliance burden emerged as the top validation challenge in 2024, replacing shortage of human resources which was the top concern in 2022-2023
- Digital transformation is accelerating: 83% of respondents are either using or planning to adopt digital validation systems. The top benefits include improved data integrity, continuous audit readiness, and global standardization.
- 79% of those using digital validation rely on third-party software providers
- Does this mean that 21% of respondents are in companies that have created their own bespoke systems? Or is something else going on there
- 63% reported that ROI from digital validation systems met or exceeded expectations
- 79% of those using digital validation rely on third-party software providers
- Artificial intelligence and machine learning are on the rise: 70% of respondents believe AI and ML will play a pivotal role in the future of validation.
- Remote audits are becoming more common: 75% of organizations conducted at least some remote regulatory audits in the past year.
- Challenges persist: The industry faces ongoing challenges in balancing costs, attracting talent, and keeping pace with technological advancements.
- 61% reported an increase in validation workload over the past 12 months
- Industry 4.0 adoption is growing: 60% of organizations are in the early stages or actively implementing Industry/Pharma 4.0 technologies.
- Digital Transformation:
As highlighted in the 2024 State of Validation report and my previous blog post on “Challenges in Validation,” several key trends and challenges are shaping the future of validation in biotech:
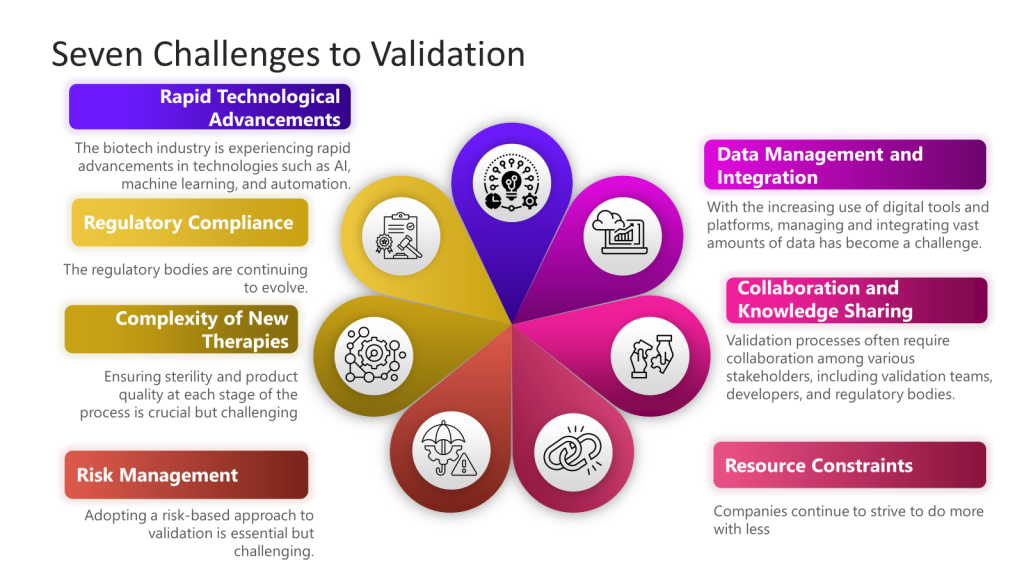
- Technological Integration: The integration of AI, machine learning, and automation into validation processes presents both opportunities and challenges. While these technologies offer the potential for increased efficiency and accuracy, they also require new validation frameworks and methodologies.
- Regulatory Compliance: Keeping pace with evolving regulatory standards remains a significant challenge. Regulatory bodies are continuously updating guidelines to address technological advancements, requiring companies to stay vigilant and adaptable.
- Data Management and Integration: With the increasing use of digital tools and platforms, managing and integrating vast amounts of data has become a critical challenge. The industry is moving towards more robust data analytics and machine learning tools to handle this data efficiently.
- Resource Constraints: Particularly for smaller biotech companies, resource limitations in funding, personnel, and expertise can hinder the implementation of advanced validation techniques.
- Risk Management: Adopting a risk-based approach to validation is essential but challenging. Companies must develop effective strategies to identify and mitigate risks throughout the product lifecycle.
- Collaboration and Knowledge Sharing: Ensuring effective communication and data sharing among various stakeholders is crucial for streamlining validation efforts and aligning goals.
- Digital Transformation: The industry is witnessing a shift from traditional, paper-heavy validation methods to more dynamic, data-driven, and digitalized processes. This transformation promises enhanced efficiency, compliance, and collaboration.
- Workforce Development: We are a heavily experience driven field. With 38% of validation professionals having 16 or more years of experience, there’s a critical need for knowledge transfer and training to equip newer entrants with necessary skills.
- Adoption of Computer Software Assurance (CSA): The industry is gradually embracing CSA processes, driven by recent FDA guidance, though there’s still considerable room for further adoption. I always find this showing up in surveys to be disappointing, as CSA is a racket, as it basically is already existing validation principles. But consultants got to consult.
- Focus on Efficiency and Audit Readiness: Creating process efficiencies and ensuring audit readiness have emerged as top priorities for validation programs.
As the validation landscape continues to evolve, it’s crucial for biotech companies to embrace these changes proactively. By leveraging new technologies, fostering collaboration, and focusing on continuous improvement, the industry can overcome these challenges and drive innovation in validation processes.
The future of validation in biotech lies in striking a balance between technological advancement and regulatory compliance, all while maintaining a focus on product quality and patient safety. As we move forward, it’s clear that the validation field will continue to be dynamic and exciting, offering numerous opportunities for innovation and growth.