Steven Shorrock writes about “Work-as-Observed” on his blog Humanistic Systems, as part of a great series on Work-as-Done. This is a nice framework for the activity of going to the Gemba, and I strongly recommend the entire series.

Steven Shorrock writes about “Work-as-Observed” on his blog Humanistic Systems, as part of a great series on Work-as-Done. This is a nice framework for the activity of going to the Gemba, and I strongly recommend the entire series.

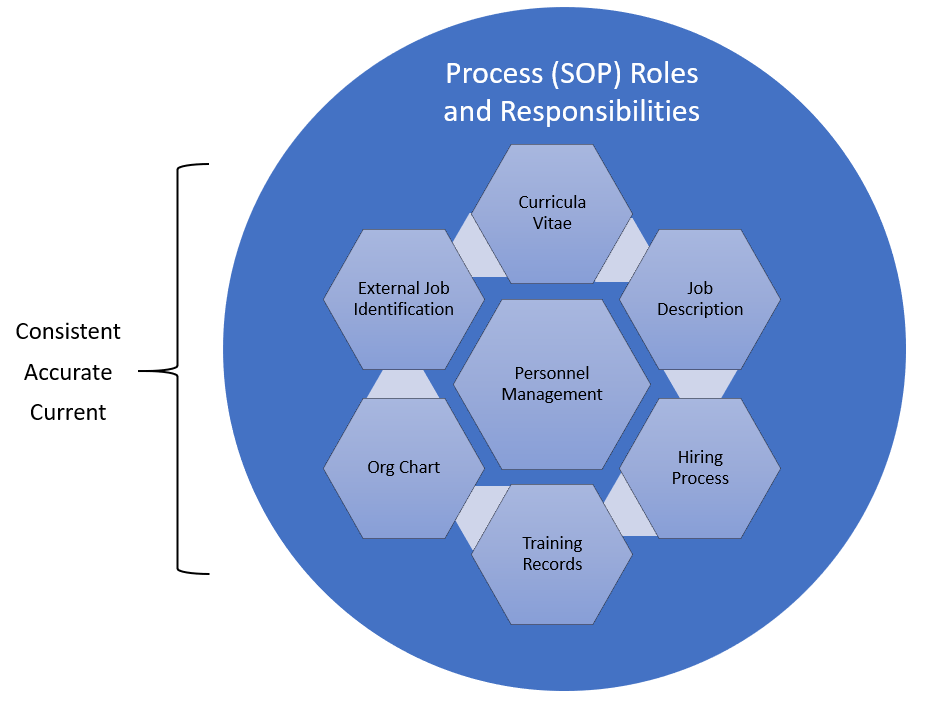
In the post “HR and Quality, joined at the hip” I covered some of the regulations that set the expectations in the pharmaceutical and medical device industries that employees have the appropriate education, experience and training to do their jobs. What is often called the personnel qualification triangle.
A job description is a written document that outlines the duties, responsibilities, contributions, behaviors, outcomes and required qualifications for a specific job in an organization. A good job description is a specification that results from a detailed job analysis. It is used in hiring and performance evaluation. It is also the starting point for determining a good GxP training plan.
In order to providing the specific knowledge, skills, abilities and behaviors that need to be addressed for each employee, the job description needs to specifically call out the roles in the processes an employee will play. Instead of providing broad statements like “participate in CAPAs” or “Manufacture product” it should be more specific such as “create and project manage CAPAs” or “Perform visual inspection.”
I challenge everyone to think broadly about the job description as a tool to drive excellence. Utilized throughout the life of employment, a job description is a powerful tool that can aid managers. Managers have a road map that can help them with their duties of planning, leading, organizing, controlling and staffing. With a road map, the complexities of the organization become easier.
The curricula vitae provides evidence that the employee is fit-for-purpose to the job description. The curricula vitae shows education and experience that demonstrates the possession of knowledge, skills, abilities and behaviors.
The training plan then lays out what the employee needs to bit fit-for-use, to be able to do all the roles in the job description. It includes all the process and role specific training, as well as filling in any gaps that might exist on the curricula vitae.
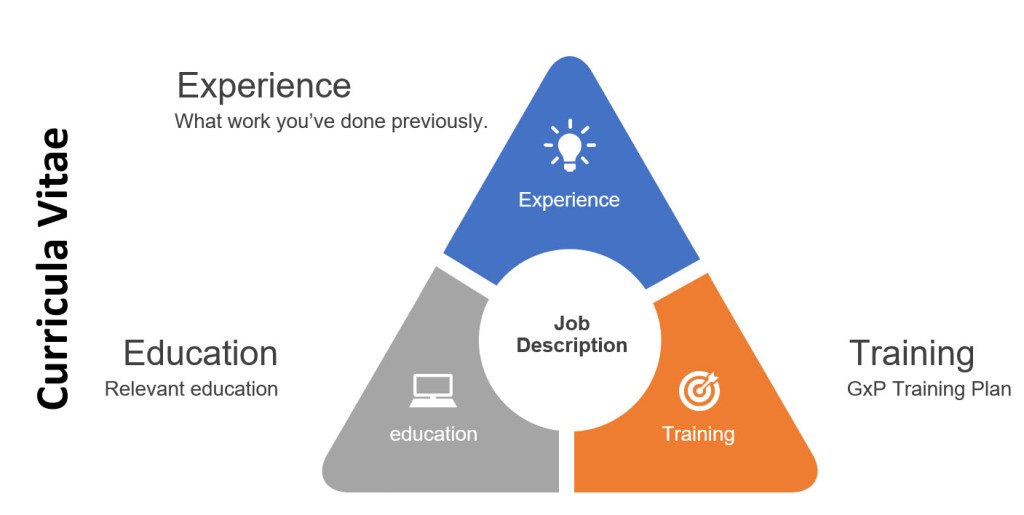
It is important to note that this may not be a fine equilateral triangle. Experience, for example, can often, but not always make up for education.
Improvement is a process and sometimes it can feel like it is a one-step-forward-two-steps-back sort of shuffle. And just like any dance, knowing the steps to avoid can be critical. Here are some important ones to consider. In many ways they can be considered an onion, we systematically can address a problem layer and then work our way to the next.
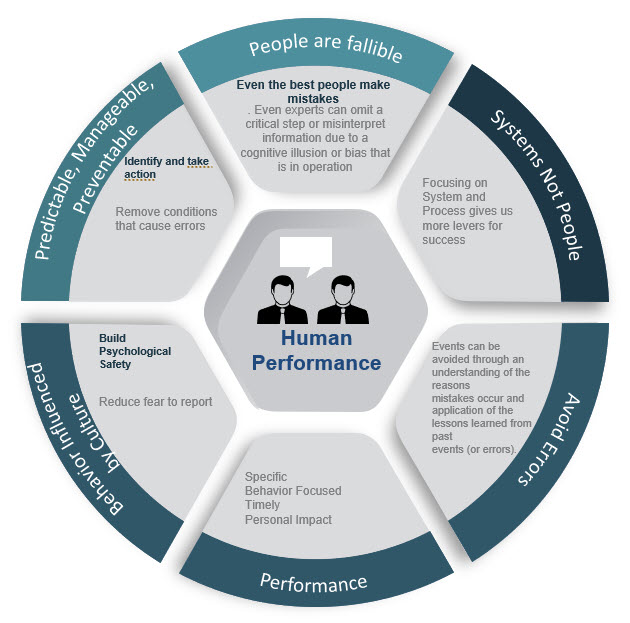
The vague, ambiguous and poorly defined bucket concept called human error is just a mess. Human error is never the root cause; it is a category, an output that needs to be understood. Why did the human error occur? Was it because the technology was difficult to use or that the procedure was confusing? Those answers are things that are “actionable”—you can address them with a corrective action.
The only action you can take when you say “human error” is to get rid of the people. As an explanation the concept it widely misused and abused.
| Attribute | Person Approach | System Approach |
| Focus | Errors and violations | Humans are fallible; errors are to be expected |
| Presumed Cause | Forgetfulness, inattention, carelessness, negligence | “Upstream” failures, error traps; organizational failures that contribute to these |
| Countermeasure to apply | Fear, more/longer procedures, retraining, disciplinary measures, shaming | Establish system defenses and barriers |
Human error has been a focus for a long time, and many companies have been building programmatic approaches to avoiding this pitfall. But we still have others to grapple with.
We like to build our domino cascades that imply a linear ordering of cause-and-effect – look no further than the ubiquitous presence of the 5-Whys. Causal chains force people to think of complex systems by reducing them when we often need to grapple with systems for their tendency towards non-linearity, temporariness of influence, and emergence.
This is where taking risk into consideration and having robust problem-solving with adaptive techniques is critical. Approach everything like a simple problem and nothing will ever get fixed. Similarly, if every problem is considered to need a full-on approach you are paralyzed. As we mature we need to have the mindset of types of problems and the ability to easily differentiate and move between them.
We remove human error, stop overly relying on causal chains – the next layer of the onion is to take a hard look at the concept of a root cause. The idea of a root cause “that, if removed, prevents recurrence” is pretty nonsensical. Novice practitioners of root cause analysis usually go right to the problem when they ask “How do I know I reached the root cause.” To which the oft-used stopping point “that management can control” is quite frankly fairly absurd. The concept encourages the idea of a single root cause, ignoring multiple, jointly necessary, contributory causes let alone causal loops, emergent, synergistic or holistic effects. The idea of a root cause is just an efficiency-thoroughness trade-off, and we are better off understanding that and applying risk thinking to deciding between efficiency and resource constraints.
Our problem solving needs to strive to drive out monolithic explanations, which act as proxies for real understanding, in the form of big ideas wrapped in simple labels. The labels are ill-defined and come in and out of fashion – poor/lack of quality culture, lack of process, human error – that tend to give some reassurance and allow the problem to be passed on and ‘managed’, for instance via training or “transformations”. And yes, maybe there is some irony in that I tend to think of the problems of problem solving in light of these ways of problem solving.
As it turns out, the reality-based, science-friendly communities and information sources many of us depend on also largely failed. We had time to prepare for this pandemic at the state, local, and household level, even if the government was terribly lagging, but we squandered it because of widespread asystemic thinking: the inability to think about complex systems and their dynamics. We faltered because of our failure to consider risk in its full context, especially when dealing with coupled risk—when multiple things can go wrong together. We were hampered by our inability to think about second- and third-order effects and by our susceptibility to scientism—the false comfort of assuming that numbers and percentages give us a solid empirical basis. We failed to understand that complex systems defy simplistic reductionism.
Zeynep Tufekci, “What Really Doomed Americas Coronovirus Response” published 24-Mar-2020 in the Atlantic
On point analysis. Hits many of the themes of this blog, including system thinking, complexity and risk and makes some excellent points that all of us in quality should be thinking deeply upon.
COVID-19 is not a black swan. Pandemics like this have been well predicted. This event is a different set of failures, that on a hopefully smaller scale most of us are unfortunately familiar with in our organizations.
I certainly didn’t break out of the mainstream narrative. I traveled in February, went to a conference and then held a small event on the 29th.
The article stresses the importance of considering the trade-offs between resilience, efficiency, and redundancy within the system, and how the second- and third-order impacts can reverberate. It’s well worth reading for the analysis of the growth of COVID-19, and more importantly our reaction to it, from a systems perspective.
We write and use procedures to help the user complete the task successfully and avoid undesired outcomes. Well-written procedures are an integral part of any organization for operation, managing risks, and continuous improvement. Effective procedures are important for the transfer of knowledge from the engineers/architects of the system to the users of the system.
Good procedures, and we are not talking format so this can be paper documents to a mixed reality guide, provide these four categories of information:
Procedures have a lifecycle through which they are developed, administered, used, reviewed, and updated. In the post “Document Management” I discussed the document management lifecycle.
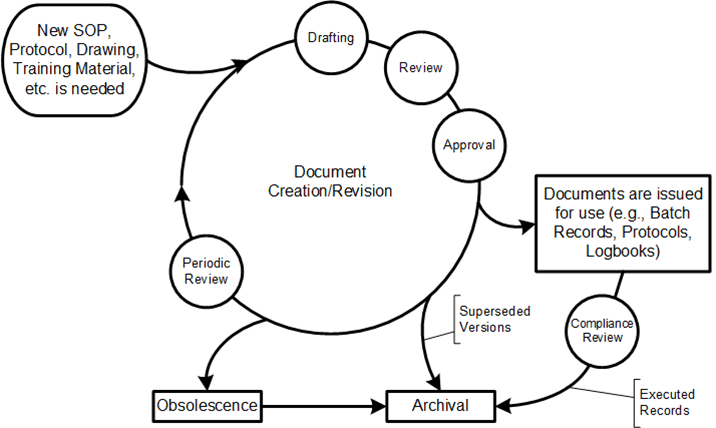
I want to focus specifically on procedures by covering five distinct phases: procedure plan, design and development, procedure authorization, procedure administration, procedure implementation and use, and procedure review and maintenance.
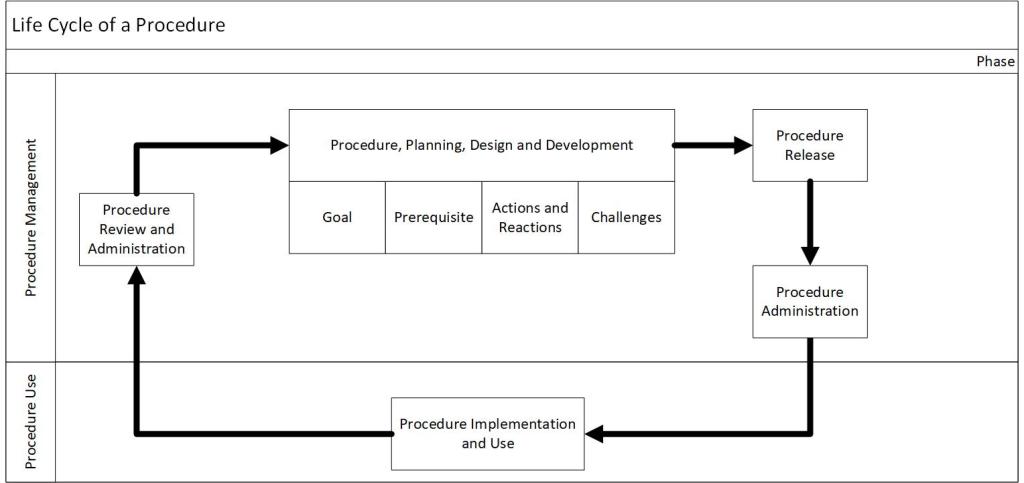
| Phase | Includes | Document Management Steps |
| Procedure plan, design and development | Identifying whether a procedure is necessary; collecting required information; producing instructions and information on the work, regulatory compliance, process and personnel safety; a walkthrough to ensure quality and potential compliance of the procedure | “New SOP is needed” Drafting |
| Procedure Authorization | Procedure review; publishing the final document; revision control; the approval process. | Review Approval |
| Procedure Administration | Managing procedure repository, control, and deployment; identifying administers how, when, and to whom procedures are to be delivered. | |
| Procedure Implementation and Use | Procedure is used in operations | |
| Procedure review and maintenance | Periodic review of documents, as well as updates from the CAPA and Change Management processes | Periodic Review |