Teams and Work-Groups need ground rules, they should have a charter, which includes a nice vision of how the team sees itself.

Let’s be honest, we do not spend enough time building and maintaining these charters. If you are like me you tend to dive right in, and that will always cause some problems. Luckily, it is never too late to take a step back and do the work.
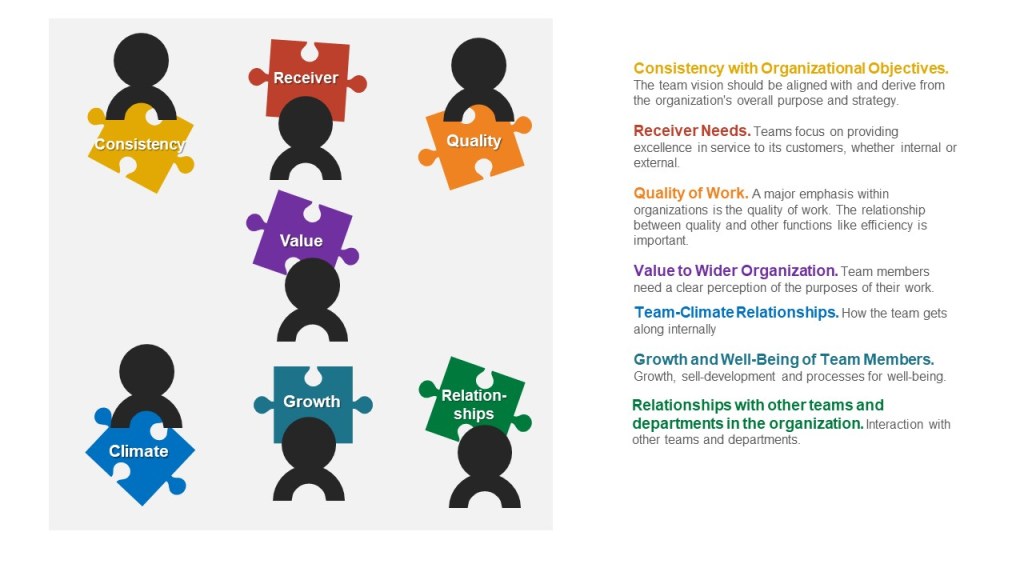
Start by answering these eight questions about the team and its place in the organization.
- Consistency with organizational objectives: The team vision should be aligned with and derive from the organization’s overall purpose and strategy. Teams are sub-elements in a wider organization structure and their success will be judged on the extent to which they make valuable contributions to the overall purpose of the organization. In some circumstances, a team may decide that it is important for its own values, purposes, and orientations to act as a minority group that aims to bring about change in organization objectives – perhaps like a red team.
- Receiver needs: How a team focuses on providing excellence in service to their customers, whether internal or external.
- Quality of work: A major emphasis within organizations is the quality of work. The relationship between quality and other functions like efficiency is important.
- Value to the wider organization: Understanding the importance of the team just not for the wider organization but beyond, leads to team cohesion and greater team effectiveness. Team members need a clear perception of the purposes of their work.
- Team-climate relationships: Team climate refers to aspects such as warmth, humor, amount of conflict, mutual support, sharing, backbiting, emphasis on status, participation, information sharing, level of criticism of each other’s work, and support for new ideas.
- Growth and well-being of team members: Growth, skill development and challenges are central elements of work-life, and teams can be a major source of support. Teams provide opportunities for skill sharing and support for new training. Teams need to be concerned for the well-being of their members, including things like burnout.
- Relationships with other teams and departments in the organization: Teams rarely operate in isolation. They interact with other team and departments within the organization. Teams must be committed to working effectively and supporting other teams. Avoid silo thinking.
From there you can then generate the 10 aspects of the charter:
- Team Ground Rules: What positive behaviors/values will the team seek to embrace and exemplify?
- Trust Damagers and Destroyers: What negative behaviors would damage/destroy trust in the team and how will the team avoid/deal with these?
- Conflicts of Interest: Are there any possible conflict of interest scenarios and, if so, how should they be handled?
- Team Boundaries: What are the boundaries of the team and the different types of team participation?
- Information Sharing: Where will there be transparency of information sharing and where will there be privacy and restricted sharing within the team?
- Issue and Conflict Resolution: How will issues and conflicts be resolved?
- Decision Making Practices: How will decisions be made?
- Meetings: Type, frequency, purpose, attendees and channels
- Induction, Mentoring, Buddying: How (and if) will new members be brought into the team?
- Communications: What tools will be used for communications and what are the agreed to “Reply-by” times
Do not short communication
How much time do you spend communicating every day? Whether this is through phone calls, IM, texts, email, written reports, face-to-face conversations, or meetings, many of us spend a large proportion of our day dealing with messages that demand our attention.
Be deliberate in how we manage these interactions so that people communicate efficiently and effectively. Outline preferred methods of communication, how to use different channels effectively, and what people want to achieve. By improving communication you can drive for effective meetings, reduce the volume of emails, ensure that exchanges are professional, and free up time for high-value tasks.
Include the following in your communication section:
- When people need to reply to emails and when they don’t.
- When people should “Reply All” to emails and when they should avoid it.
- How to organize regular team meetings, who should attend, whether people will “dial in” remotely, what to include in the minutes, who will circulate them, and so on.
- How your team communicates with customers internal and external.
- How team members interact on internal social media (chat, slack, etc).
- When making a video or audio call is appropriate.
- How people engage with others face-to-face.
Signs of Team/Work-Group Misalignment
There are some behaviors to look out for, when you see them, it’s time to return to the charter, and improve.
Vague Feelings of Fear. You know what the team is supposed to deliver on, but you don’t know how exactly you’re supposed to work with anything in your power or control to “move the needle.”
Ivory Tower Syndrome. Things aren’t clear or different people have different expectations for a project or initiative. No one is really able to clarify.
Surprises. Someone committed you to a task, but you weren’t part of that decision.
Emergencies. How often are you called on to respond to something that’s absolutely needed by close of business today? How often are you expected to drop everything and take care of it? How often do you have to work nights and weekends to make sure you don’t fall behind?
Cut Off at the Pass: Someone else is doing the same work unaware to all.
Not Writing Things Down. You have to make sure everyone is literally on the same page, seeing the world in a similar enough way to know they are pursuing the same goals and objectives. If you don’t write things down, you may be at the mercy of cognitive biases later. How do you know that your goals and objectives are aligned with your overall company strategy? Can you review written minutes after key meetings? Are your team’s strategic initiatives written and agreed to by decision-makers? Do you implement project charters that all stakeholders have to sign off on before work can commence? What practices do you use to get everyone on the same page?