Microbiologists won’t be sequestered in the laboratory, running samples and conducting environmental testing, once the revisions proposed for Annex 1 of the EU and Pharmaceutical Inspection Cooperation Scheme (PIC/S) GMP guides take effect, Annex 1 rapporteur Andrew Hopkins said Oct. 15.
They will have a broader role that includes conducting risk assessments to ensure that sterile products are made as contamination-free as possible, said Hopkins, who is an inspector for the UK Medicines and Healthcare products Regulatory Agency.
Pink Sheet “EU GMP Annex 1 Would Give Microbiologists A Greater Role In Sterility Assurance, Rapporteur Says“
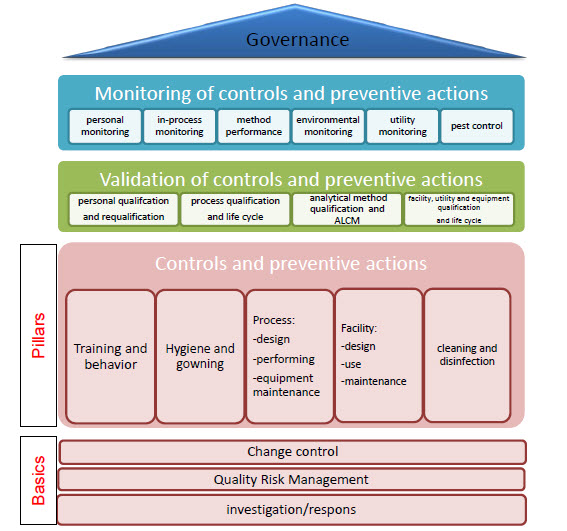
Contamination Control is a fairly wide term used to mean “getting microbiologists out of the lab” and involved in risk management and compliance. Our organization splits that function off from the QC Microbiology organization but there are many models for making it work.
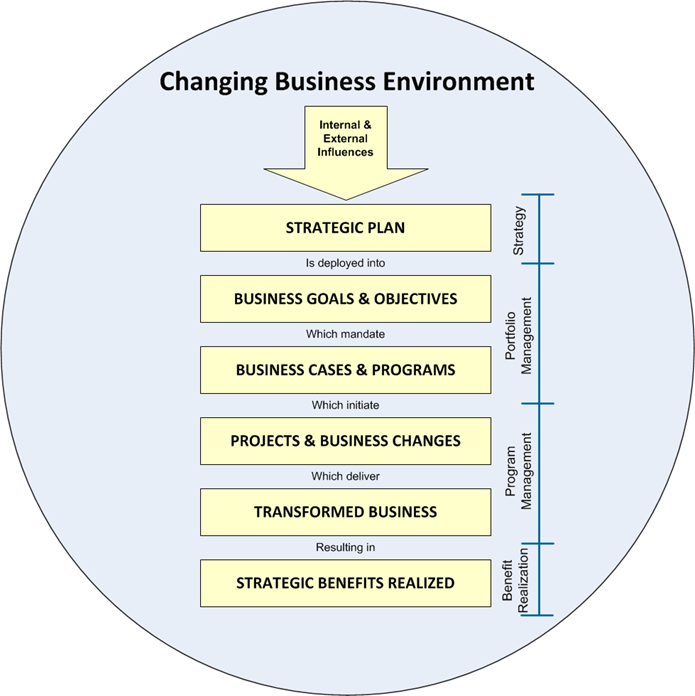
Risk Management is a major part of the new Annex 1, and what they are driving at are good risk assessments with good risk mitigation that involve the microbiologists.
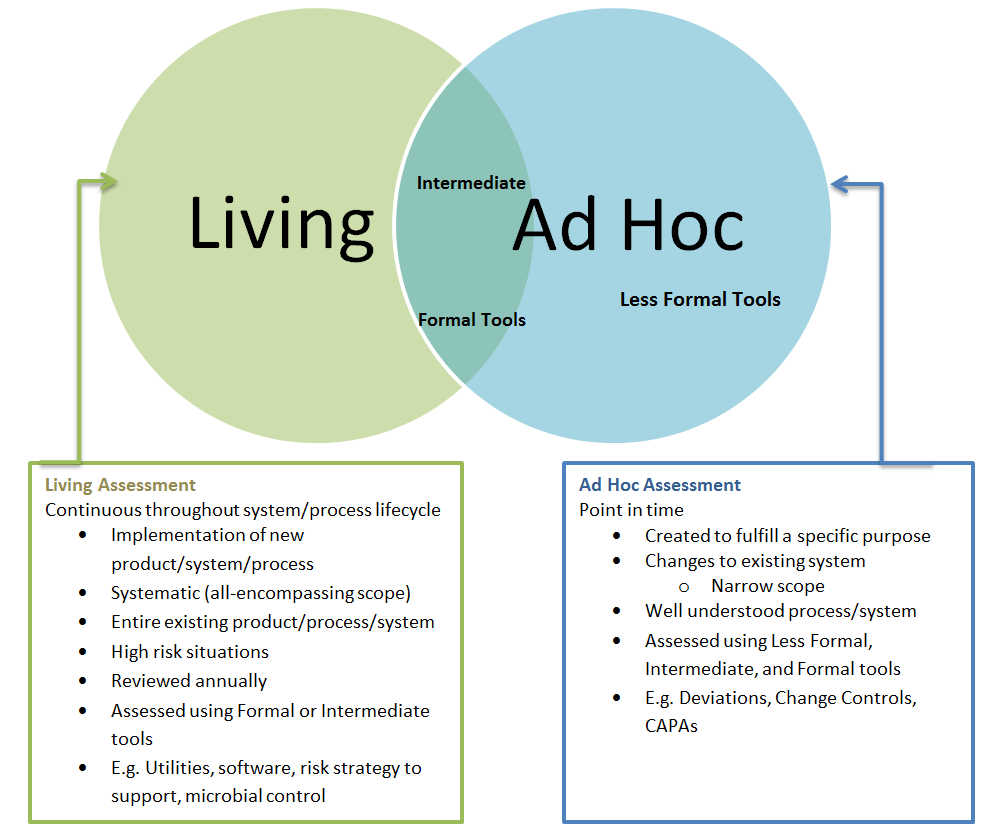
This is really what is meant by a contamination control strategy which considers the product and process knowledge and skills in pharmaceutical product manufacturing and GMP/ cGMP compliance under the auspices of a Pharmaceutical Quality System (Q10) together with initiatives of Quality by Design (Q8) and Quality Risk Management (Q9).
From this strategy comes:
- Targeted/ risk based measures of contamination avoidance
- Key performance indicators to assess status of contamination control
- A defined strategy for deviation management (investigations) and CAPA
When it comes to change management, one of the easiest places to go wrong is to forget to bring the microbiologist in to changes. Based on your strategy you can determine change changes require their assessment and include it in the tool utilized to determine SMEs, for example:
| Department | Required if the change meets any of the following criteria: |
| Contamination Control | The change impacts environment integrity, conditions or monitoring, including:
The change can impact microbiological control within a process stream, raw material or process equipment The changes are to water systems |