Project Management, the structured approach for managing tasks, resources, and budget to achieve a defined deliverable, is an important part of the quality management toolbox, and an important aspect to build into your change management program.
There are a lot of project management methodologies, but they all boil down to having an understanding of the tradeoffs between time, cost, and scope of a change; and then motivating a team towards delivering the change.
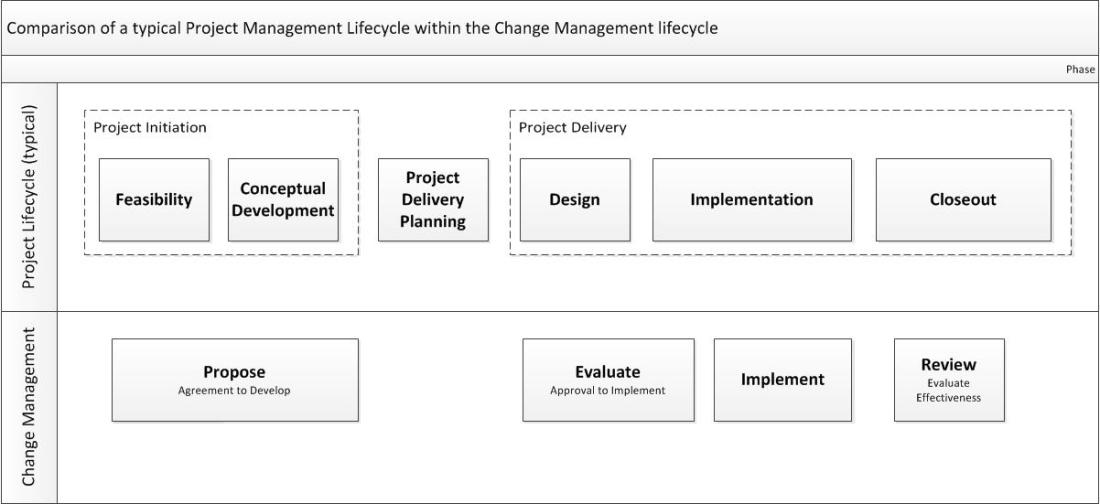
Evaluate is best seen as a gate at the end of the project’s design (or a phase of design). A well-designed project, with appropriate stakeholders and team members ideally, will flow nicely into an evaluate change control (or set of change controls). Having this as part of your gate to develop will ensure that the right subject-matter-experts have been involved, that all potential risks and impacts are understood, and will ensure the site is ready to implement.
Small projects and the change management lifecycle are usually one and the same.
For projects with a large impact, it is often important to create more than one change control, to ensure appropriate implementations. In these cases it is often useful to create a change strategy (or incorporate in a project deliverable, such as a gate document) which can include:
| Scope | Describe the project, indicating the current and future state. |
| Roles/ Responsibilities | Indicate what functions/departments will serve what role in the change controls
Indicate the role and responsibilities of project management. |
| Methodology/ Strategy | What activities are included and how they will be organized.
How many change controls will be created as part of this strategy. How change controls will be organized and linked together (e.g. dependencies). Provide a methodology for managing changes and ensuring all change control activities happen according to the provided timetable. Things to Consider: Are there other changes that affect the same room/area or equipment? How are affected rooms/areas/equipment being taken out of and placed back into service as to not to interfere with the provided timetable? Will these change controls affect daily operations/sampling? How will changes be organized? For example, will changes be organized along install, validate and implement? If multiple areas are to be affected, will there be changes in Material and Personnel Flow that will need to be modified to execute the multiple changes? |
| Effectiveness Review | If the project will have one effectiveness review (e.g. a process validation, comparability protocol, stability study) indicate, provide the effectiveness review criteria and justify. Provide a timeline for completion of the effectiveness review. |
| Regulatory Strategy | Regulatory strategy for filing the project (e.g. will the changes be filed independently or together, at what point, which changes will require regulatory assessment) |
| Planned Timetable | Timelines for writing, approving, implementing and closing change controls.
Take the dependencies written in the Methodology/Strategy into consideration when developing the timetable |
| Closure Plan | How will the strategy be closed? What are the criteria for a successful project closure? |
For larger projects, the change evaluation will start in the project design phase but can continue through implementation as individual changes are put in place.
The advantage of writing this strategy allows the project to consolidate deliverables and ensure the right level of effort is put into the changes across the project.