A month back on LinkedIn I complained about a professional society pushing the idea of a document-free quality management system. This has got to be one of my favorite pet peeves that come from Industry 4.0 proponents, and it demonstrates a fundamental failure to understand core concepts. And frankly one of the reasons why many Industry/Quality/Pharma 4.0 initiatives truly fail to deliver. Unfortunately, I didn’t follow through with my idea of proposing a session to that conference, so instead here are my thoughts.
Fundamentally, documents are the lifeblood of an organization. But paper is not. This is where folks get confused. But fundamentally, this confusion is also limiting us.
Let’s go back to basics, which I covered in my 2018 post on document management.
When talking about documents, we really should talk about function and not just by name or type. This allows us to think more broadly about our documents and how they function as the lifeblood.
There are three types of documents:
- Functional Documents provide instructions so people can perform tasks and make decisions safely effectively, compliantly, and consistently. This usually includes things like procedures, process instructions, protocols, methods, and specifications. Many of these need some sort of training decision. Functional documents should involve a process to ensure they are up-to-date, especially in relation to current practices and relevant standards (periodic review)
- Records provide evidence that actions were taken, and decisions were made in keeping with procedures. This includes batch manufacturing records, logbooks and laboratory data sheets and notebooks. Records are a popular target for electronic alternatives.
- Reports provide specific information on a particular topic on a formal, standardized way. Reports may include data summaries, findings, and actions to be taken.
The beating heart of our quality system brings us from functional to record to reports in a cycle of continuous improvement.
Functional documents are how we realize requirements, that is the needs and expectations of our organization. There are multiple ways to serve up the functional documents, the big three being paper, paper-on-glass, and some sort of execution system. That last, an execution system, united function with record, which is a big chunk of the promise of an execution system.
The maturation mind is to go from mostly paper execution, to paper-on-glass, to end-to-end integration and execution to drive up reliability and drive out error. But at the heart, we still have functional documents, records, and reports. Paper goes, but the document is there.
So how is this failing us?
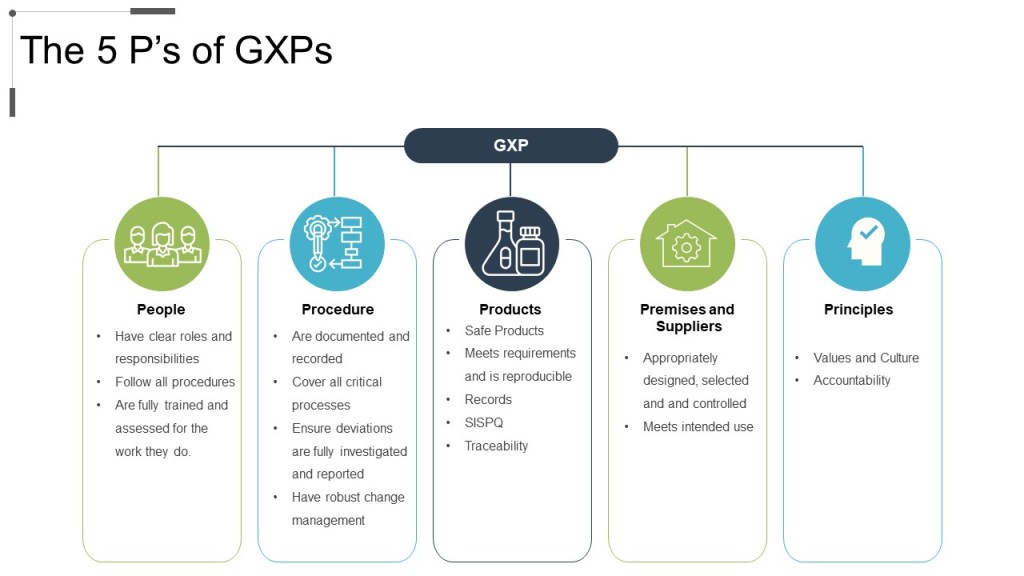
Any process is a way to realize a set of requirements. Those requirements come from external (regulations, standards, etc) and internal (efficiency, business needs) sources. We then meet those requirements through People, Procedure, Principles, and Technology. They are interlinked and strive to deliver efficiency, effectiveness, and excellence.
So this failure to understand documents means we think we can solve this through a single technology application. an eQMS will solve problems in quality events, a LIMS for the lab, an MES for manufacturing. Each of these is a lever for change but alone cannot drive the results we want.
Because of the limitations of this thought process we get systems designed for yesterday’s problems, instead of thinking through towards tomorrow.
We get documentation systems that think of functional documents pretty much the same way we thought of them 30 years ago, as discrete things. These discrete things then interact through a gap with our electronic systems. There is little traceability, which complicates change control and makes it difficult to train experts. The funny thing, is we have the pieces, but because of the limitations of our technology we aren’t leveraging them.
The v-model approach should be leveraged in a risk-based manner to the design of our full system, and not just our technical aspects.

System feasibility matches policy and governance, user requirements allow us to trace to what elements are people, procedure, principles, and/or technology. Everything then stems from there.