
Ah, the New Year. A time of trying to remember how to write the correct date and mostly failing. A time where I practice my capability to make simple corrections on forms still completed by hand.

Ah, the New Year. A time of trying to remember how to write the correct date and mostly failing. A time where I practice my capability to make simple corrections on forms still completed by hand.
I spend a lot of time discussing uncertainty and how to address it in our quality system and within our organization. However, we often find ourselves at a crossroads, faced with uncertainty and the unknown in our careers – certainly, the last few years have been hard in biotech. My current approach has been to reframe this uncertainty not as an obstacle but as a feature of my journey—something it might have taken me 54 years to learn. I am striving to embrace the concept of “trusting the process” personally and as a quality practitioner so I can navigate life’s twists and turns with greater ease and purpose. As we go into the New Year, here are my current approaches.
If you are like me, it is easy to get lost in the day-to-day pressures of work. There is always a new issue, a new course correction. It is easy to focus on the overwhelming big picture to our next best steps and forget that the journey counts. My QA problem-solving self often wants to focus on problem-solving and forgets that we must strike a balance between action and acceptance, recognizing that while we can’t control every outcome, we can control our response to each situation. I am working to maintain agency in the present moment while surrendering to the unfolding path ahead.
Uncertainty, often viewed as a source of anxiety, can actually be a powerful catalyst for growth and innovation. By reframing uncertainty as a feature, we can open ourselves up to new possibilities and unexpected opportunities. This mindset shift encourages us to:
One practical way to embrace uncertainty is through the practice of running small experiments. These controlled tests allow us to:
We create a culture of continuous learning and improvement by incorporating regular experimentation into our personal and professional lives. This approach is particularly valuable when balancing the demands of serving an organization while pursuing personal growth.
The challenge of running small experiments while fulfilling organizational responsibilities is common. Here are some strategies to help strike that balance:
Trusting the journey is not about blind faith or passivity. Instead, it’s about developing a deep relationship with your wisdom and decision-making process. This trust is built over time through:
As you cultivate this trust, you’ll find yourself better equipped to navigate uncertainty confidently and gracefully.
Trusting the journey can feel counterintuitive in a world that often demands certainty and immediate results. However, by embracing uncertainty as a feature of our growth process, we open ourselves to a richer, more fulfilling experience. Through small experiments, mindful action, and a willingness to surrender to the unknown, we can create a life and career that is both purposeful and adaptable.
Remember, the journey itself is where true growth and discovery happen. By trusting the process and focusing on our next best steps, we can navigate the complexities of life with greater ease and authenticity. So, take that first step, run that small experiment, and trust that the journey will unfold in ways you may never have imagined.
This is my New Year’s plan: to continue to apply to my personal space the skills and mindsets that have made my career so fruitful.
Defining a GMP critical system is an essential aspect of Good Manufacturing Practices (GMP) in the pharmaceutical and medical device industries. A critical system is one that has a direct impact on product quality, safety, and efficacy.
It’s important to note that the criticality of systems should be periodically evaluated to ensure they remain in a valid state and compliant with GMP requirements. This includes reviewing the current range of functionality, deviation records, incidents, problems, upgrade history, performance, reliability, security, and validation status reports.
The FDA has published a draft guidance for “Protocol Deviations for Clinical Investigations of Drugs, Biological Products, and Devices.”
This draft guidance adopts the ICH E3(R1) definitions for protocol deviation and important protocol deviation, providing more standardized terminology, which is a great thing. Avoiding the term “protocol violation”, it primarily uses “protocol deviation” and “important protocol deviation.”
The FDA guidance provides detailed sections on the roles and responsibilities of investigators, sponsors, and IRBs in monitoring, mitigating, and reporting protocol deviations. It as specific recommendations for reporting protocol deviations to sponsors, IRBs, and FDA, including timelines and methods.
It mostly seems a good application of a quality-by-design approach, focusing on critical-to-quality factors and risk-based monitoring for clinical studies. Hopefully it will help clear up confusion in this area.
Effective risk analysis is crucial for informed decision-making and robust risk management. Simply conducting a risk analysis is not enough; its effectiveness in engaging the risk management decision-making process is paramount. This effectiveness is largely driven by the transparency and documentation of the analysis, which supports both stakeholder and third-party reviews. Let’s explore how we can measure this effectiveness and why it matters.
Transparency and documentation form the backbone of an effective risk analysis process. They ensure that the methodology, assumptions, and results of the analysis are clear and accessible to all relevant parties. This clarity is essential for:
To gauge the effectiveness of risk analysis in engaging the decision-making process, consider the following metrics:
Measure the degree to which stakeholders actively participate in the risk analysis process and utilize its outputs. This can be quantified by:
Assess how often risk analysis findings directly influence management decisions. Track:
Evaluate the clarity and comprehensiveness of risk reports. Consider:
This is a great place to leverage a rubric.
Analyze the results of internal and external audits or reviews:
Measure how frequently risk analysis tools and outputs are accessed and used:
To implement these metrics effectively:
To improve the effectiveness of risk analysis through better transparency and documentation:
Develop standardized templates and formats for risk reports to ensure consistency and completeness. This standardization facilitates easier comparison and analysis across different time periods or business units.
Create a common language for risk across the organization. A well-defined risk taxonomy ensures that all stakeholders understand and interpret risk information consistently.
Utilize data visualization techniques to present risk information in an easily digestible format. Visual representations can make complex risk data more accessible to a broader audience, enhancing engagement in the decision-making process.
Document all steps of the risk analysis process, including data sources, methodologies, assumptions, and decision rationales. This audit trail is crucial for both internal reviews and external audits.
Encourage open communication about risks throughout the organization. This cultural shift can lead to more honest and accurate risk reporting, ultimately improving the quality of risk analysis.
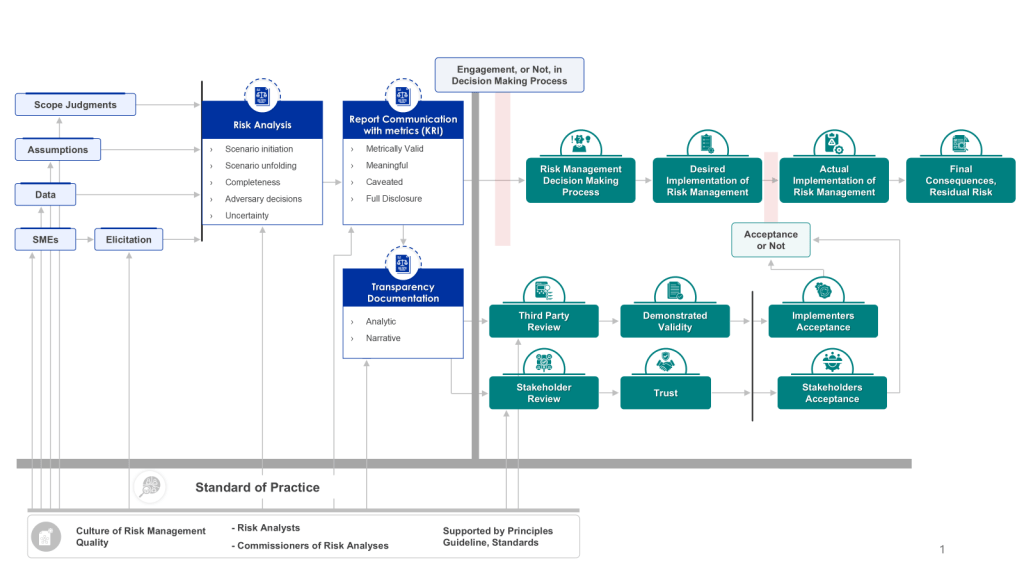
Measuring the effectiveness of risk analysis in engaging the risk management decision-making process is crucial for organizations seeking to optimize their risk management strategies. By focusing on transparency and documentation, and implementing key metrics to track performance, organizations can ensure that their risk analysis efforts truly drive informed decision-making and robust risk management.
Remember, the goal is not just to conduct risk analysis, but to make it an integral part of the organization’s decision-making fabric. By continuously measuring and improving the effectiveness of risk analysis, organizations can build resilience, enhance stakeholder trust, and navigate uncertainties with greater confidence.