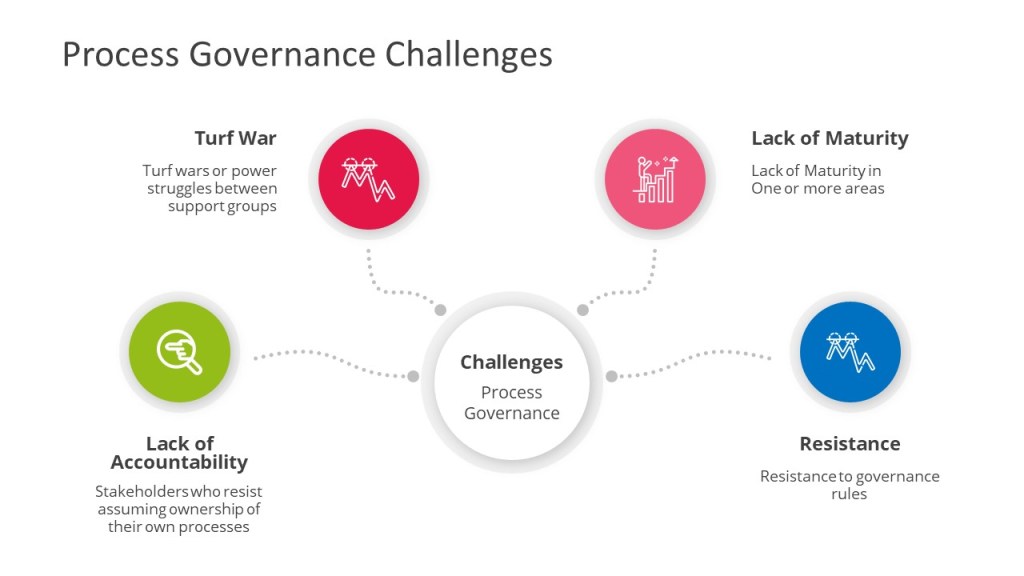
Governance and ownership challenges often arise in an organization for four reasons:
- Business stakeholders who resist assuming ownership of their own processes, data and/or knowledge, or have balkanized/siloed accountability
- Turf wars or power struggles between groups of stakeholders
- Lack of maturity in one or more areas
- Resistance to established governance rules
The Business Struggles with Accountability
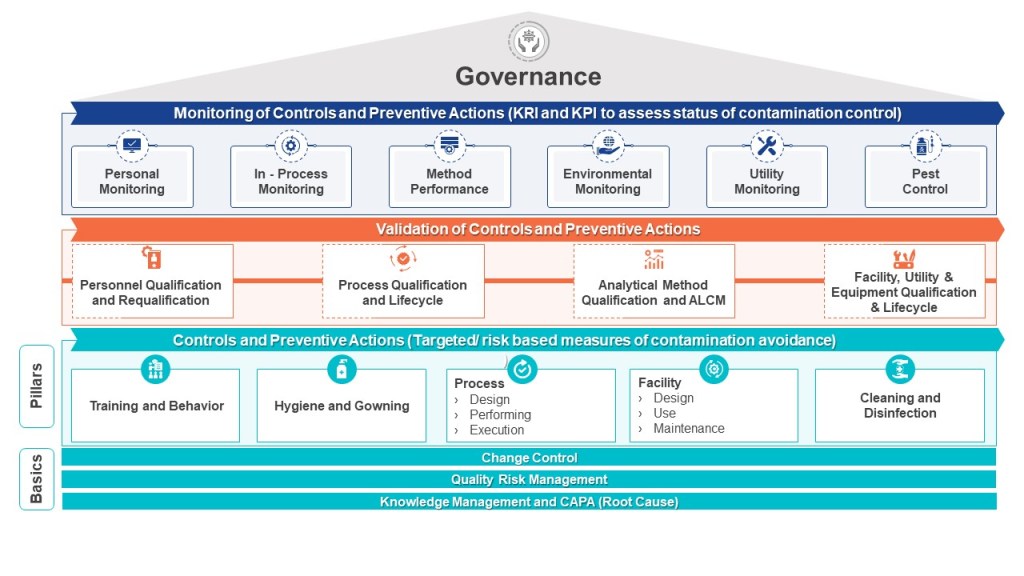
Processes often have a number of stakeholders, but no apparent owners. This results in opportunity costs as compulsory process changes (e.g. legislative requirements, systems capacity, or company structural changes) or process improvements are not implemented because the business process owner is unaware of the change, or no clear business process owner has been identified which leads to an increase in risk.
Sometimes processes have a number of stakeholders who all think they are the owners of parts of the process or the whole process. When this overlap happens, each supposed owner often identifies their own strategy for the process and issues their own process change instructions to conform to their understanding of the purpose of the business process. These conflicting instructions lead to frustration and confusion by all parties involved.
Lack of accountability in process and system leads to inefficient processes, organizational disharmony, and wasted energy that can be better spent on process improvements.
Turf Wars
Due to silo thinking there can be subdivided processes, owned by different parts of the organization. For example, count how many types of change control your organization has. This requires silos to be broken down, and this takes time.
Lack of Maturity
Governance is challenging if process maturity is uneven across the organization.
Failure to Adhere to Governance
It can be hard to get the business to apply policy and standard consistently.