The FDA has posted the 2019 483 observations as an excel file. The FDA has made these files available every year since 2006 and I find them to be one of my favorite tools for evaluating regulatory trends.
So for example, looking at change related 483 I see:
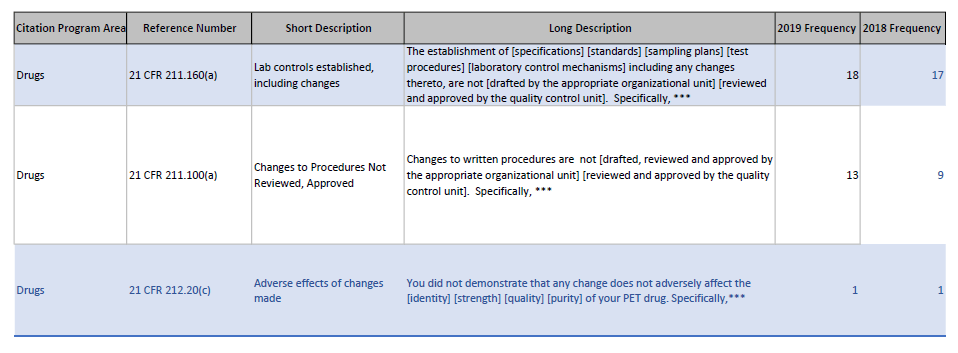
Or for data integrity issues:
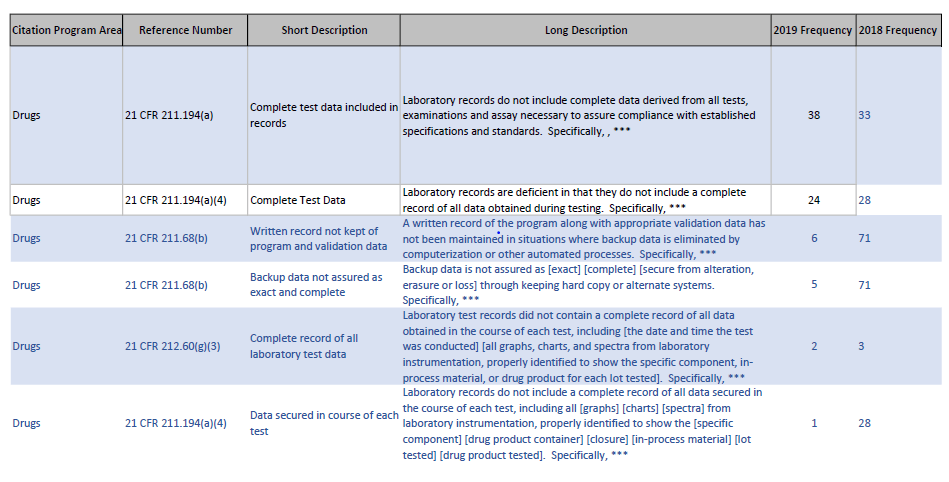
Very useful resource that should be in the bookmarks for every pharmaceutical quality professional.

2 thoughts on “FDA 483 data”