Luis Charles Chavarría recently posted about watches and quality. Reading that, and several of the responses remind how pharmaceutical quality is often just framed in terms of regulatory adherence instead of a broader approach. ISO9001 and other major quality models basically break down to having 8 dimensions of quality.
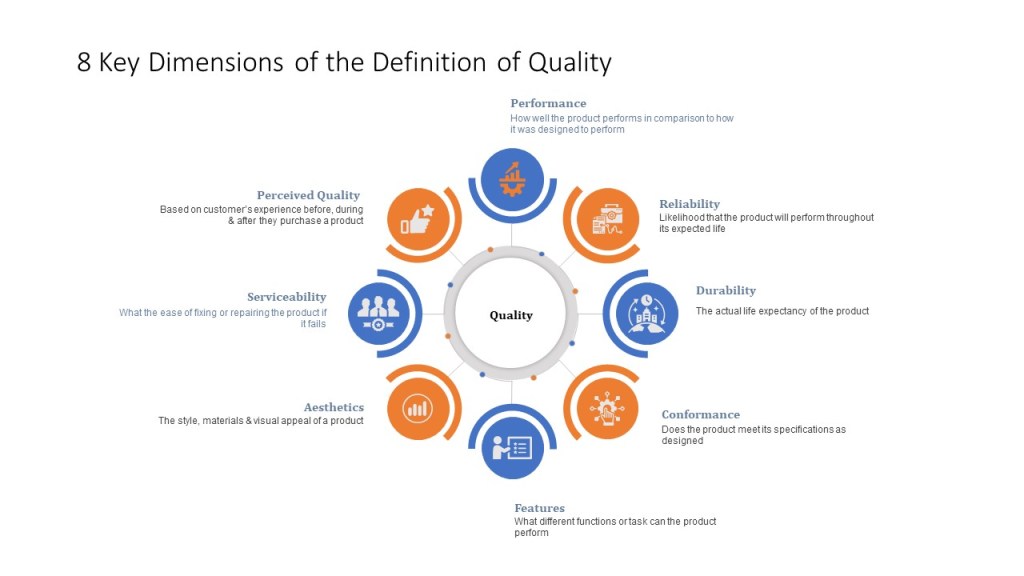
In a lot of industries, it is very visible to the customer whether quality exists. Shoes, cars, toaster ovens – I can gauge them based on multiple of the criteria above before I buy. I can go on credible review sites, use tools like Consumer Reports, get reliable feedback from others. When a friend recommends a couch, I can trust their opinions.
We have none of that in the regulated areas of pharma and much of medical devices. Patients are prescribed product, and even when there are multiple generics available the only real criteria is price. Chances are the patient barely knows the manufacturer, let alone the manufacturing site. It can be very difficult for a patient, or even doctor, to gauge the quality of the product.
It is for this reason we need more transparency throughout the supply chain, through the development of products. The National Academies of Science, Engineering and Medicine (NASEM) report is a start and needs to be implemented quickly. As a beginning it can really help start to shift the needle and make large parts of this industry more receptive to