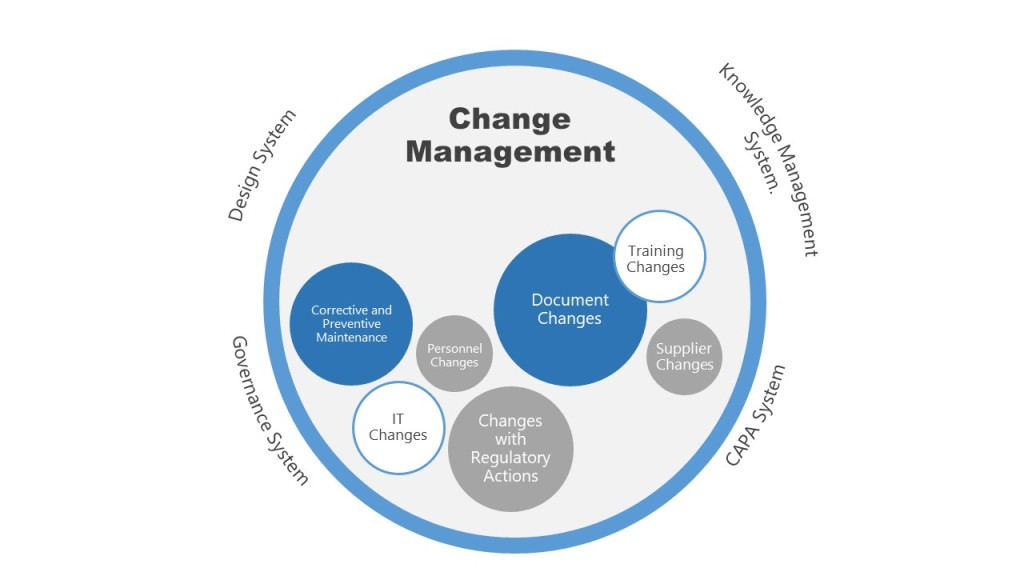
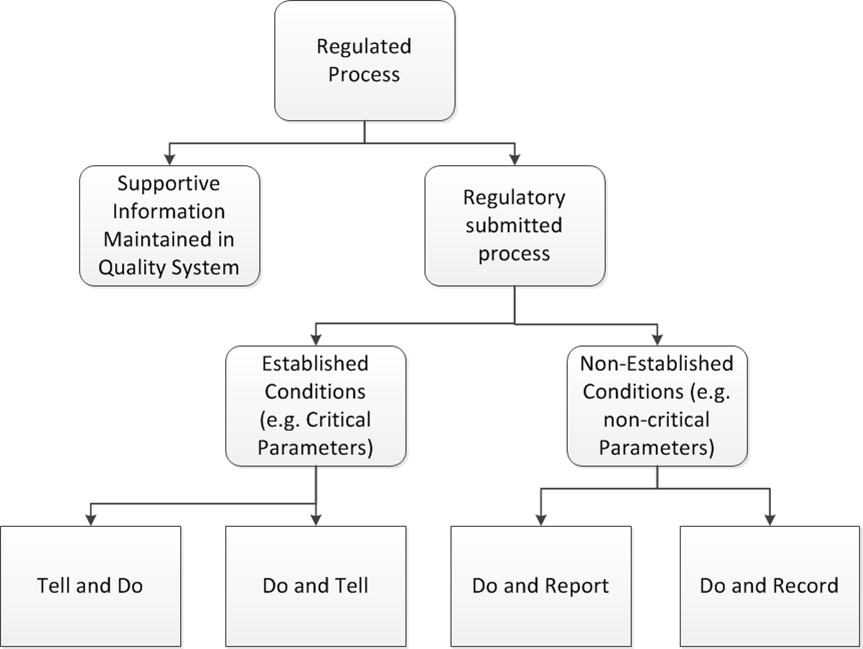
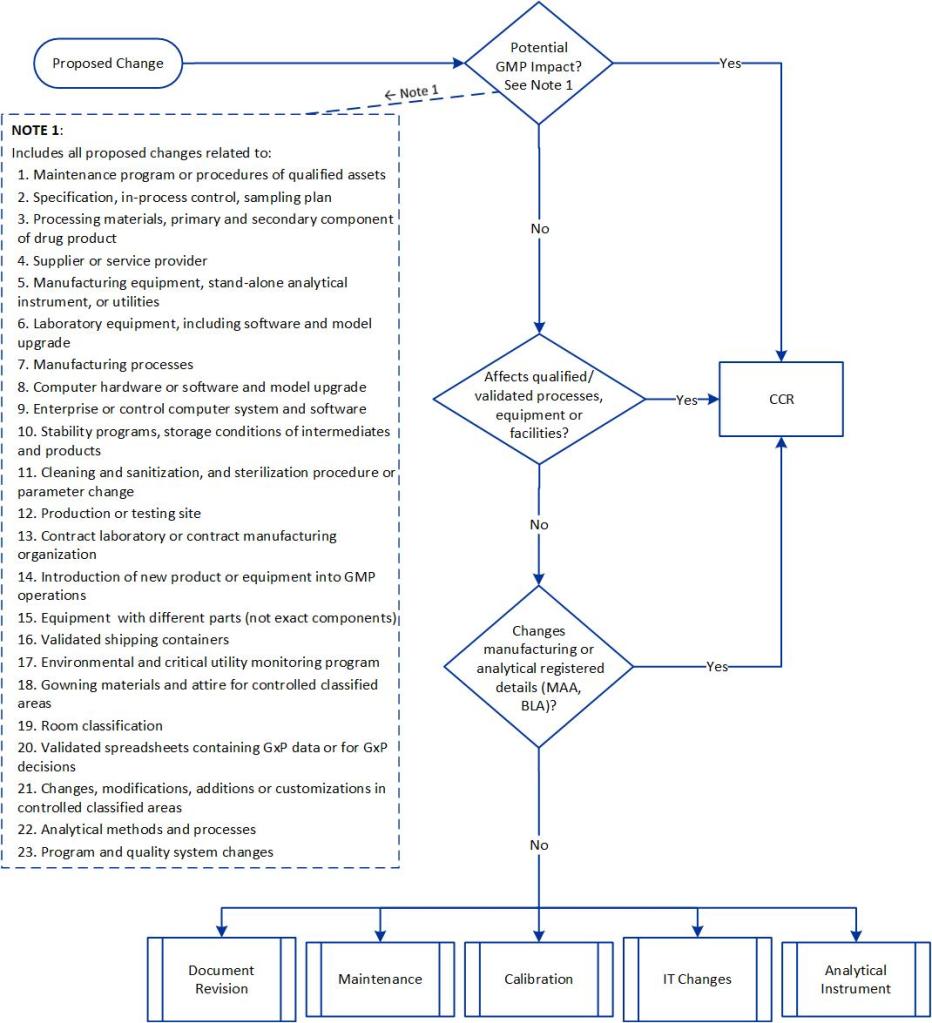
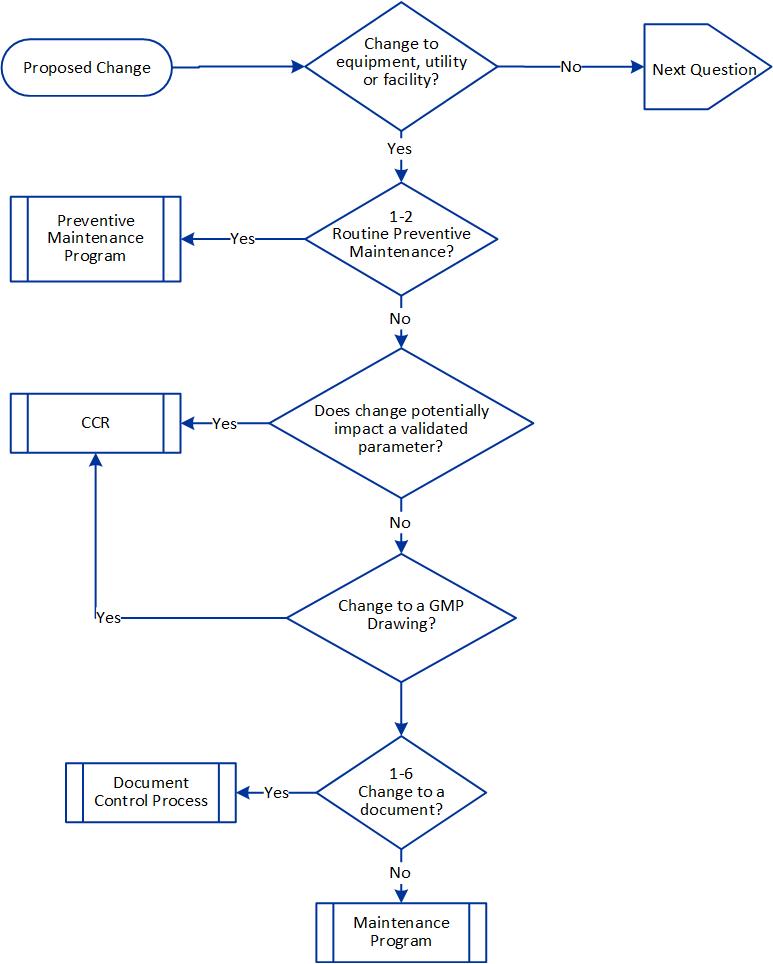
With the current plan to start attending conferences again this spring, I’ve been working a lot on a few different presentations, which means spending a lot of time on PowerPoint presentations.
Microsoft debuted PowerPoint in 1987, and since then, it has been used to present content in meetings, conference rooms, and classrooms. There are a lot of jokes about how bad PowerPoint can be, but if you know a little about its features, PowerPoint can be so much more than mere presentation software. It can be the means for taking audiences on a truly engaging learning adventure as well as a powerful tool that supports presenters by serving as their digital co-facilitator. It just requires some work.
Making presentations for folks outside my organization always gets me thinking of best practices. It helps me concentrate on how the true value of PowerPoint isn’t to serve as an information provider—that’s the role of a presenter. The true value of PowerPoint
is to support you and your presentation.
A presentation is most effective when it is focused and has a coherent narrative. Achieving that starts with defining your objectives and then taking some time to figure out how you’ll meet those objectives. Be intentional in your use of PowerPoint.
| Traditional PowerPoint Thinking | Intentional PowerPoint Design Thinking |
| Every presentation needs slides. | My intended presentation outcomes should dictate the types of visual aids I use (or don’t use). |
| Every point I make needs a slide. | My slides should never compete with me for the audience’s attention; they should support my message. |
| PowerPoint is synonymous with your presentation. | PowerPoint is my co-facilitator. |
| PowerPoint is linear, and slides appear sequentially. | Using triggers and hyperlinks, it’s possible to reveal information dynamically. |
| Templates make a slide deck look professional. | Effective use of slide real estate and visual representation of my message looks professional. |
| There is a maximum number of words and an ideal font size for most presentations. | My audience should be able to read all the words that appear on a slide. |
| People need a lot of information on technical slides and data-driven presentations. | Slides are a visual aid for a presentation; more detailed information is better offered through handouts. |
| There are lots of options for animations and transitions, so they should all be used at some point. | Animations and transitions can help focus attention, but there is such a thing as too much. |
| I can send someone my PowerPoint deck and that should be the equivalent of attending my presentation. | Most narratives can be placed in the Notes section and distributed, along with my slides, to paint a complete picture for those not in attendance. |
Give a lot of thought to who is your audience. It is always a good idea to understand your audience, but when speaking to folks outside of your What (if anything) does the potential audience already know about your topic? What should the audience be able to do new, different, or better because of the time spent with you?
For example, at the upcoming ISPE Asceptic Conference, my audience understands pharmaceutical quality systems so I can start with the understanding that they understand the basics of my topic. My presentation, as a result, can go to more advanced topics and not have to explain the basics.
A presentation is most effective when it is focused and has a coherent narrative. Achieving that starts with defining your objectives and then taking some time to figure out how you’ll meet those objectives. Taking an hour or two to map out your thoughts and truly think through how best to visually represent your key points can help ensure that your presentation will be tight and focused with a coherent flow.
For each slide:
- Slide Purpose/Objective
- Sketch/Imagery
- Key Points
Things Conferences Should Change
Working on presentations for conferences again really reminds me of all the bad practices conferences continue to use.
- Stop Using Templates: It is a common misconception is that using a template makes the slide deck look more professional. Slide templates do help with consistency, but they dramatically reduce the real estate you have to work with on your slide. By the very nature of their structure, these templates encourage a title and bulleted list format. Don’t just believe me, watch this fun video by Will Thalheimer. The more space on a slide that is occupied by professional-looking template designs and logos, the less space remains for inserting powerful imagery, text, facts, or figures.
- Leverage Technology to Break Linearity: Most people use PowerPoint in linerar ways, and conference technology builds pretty much make that an inevitability. The technology exists to allow the audience to have some sort of control over the content that’s on display in front of them, and would greatly enhance the conference experience.
Resources
- Bozarth, J. 2013. Better Than Bullet Points: Creating Engaging e-Learning with PowerPoint. 2nd ed. San Francisco: John Wiley & Sons.
- Duarte, N. 2010. Resonate: Present Visual Stories that Transform Audiences. Hoboken, NJ: John Wiley & Sons.
- Duarte, N. 2008. slide:ology: The Art and Science of Creating Great Presentations. Sebastopol, CA: O’Reilly Media.
- Medina, J. 2014. Brain Rules (2nd ed.). Seattle: Pear Press.
- Schwertly, S. 2011. How to be a Presentation God: Build, Design and Deliver Presentations that Dominate. Hoboken, NJ: John Wiley & Sons.
- Vella, J. 2002. Learning to Listen, Learning to Teach: The Power of Dialogue in Educating Adults. San Francisco: Jossey-Bass.
- Williams, R. 2008. The Non-Designer’s Design Book. 3rd ed. Berkeley, CA: Peachpit Press.